Laboratory of Virology
KEYWORDS
- Elucidation of the molecular basis of viral pathogenicity
HEAD

LAB MEMBER
| Faculty | Position | Researchers |
|---|---|---|
| MIYAKE Yasuyuki | Associate Professor | Researchers |
| WATANABE Takahiro | Lecturer | Researchers |
| SUGIMOTO Atsuko | Assistant Professor | Researchers |
CONTACT
| iga-ryu◎t.mail.nagoya-u.ac.jp (Please send a message after replacing "◎" mark with "@" mark. ) | |
| HP | Private Page |
OUTLINE
Our laboratory has been engaging in basic molecular biology, translational research, and applied research of viruses, mainly focusing on two human herpesviruses, Epstein-Barr virus (EBV) and herpes simplex virus (HSV). EBV is a member of the gamma-herpesvirus, associated with infectious mononucleosis and various types of malignancy, such as Burkitt lymphoma, Hodgkin lymphoma, post-transplant lymphoproliferative disorder, T/NK lymphoma, gastric carcinoma, and nasopharyngeal carcinoma.
Carcinogenesis of EBV-positive cancers is a multistep process. We are suggesting three common features that contribute to the development and maintenance of EBV-positive cancers: effects of EBV oncogenes, immunosuppression and evasion/exploitation of the immune system, and genetic and epigenetic predisposition/alteration of the host genome (Murata, Sato, Kimura, Rev Med Virol 2014).
We here introduce one of our recent research topics, focusing on EBV life cycle. The EBV has two alternative life cycles: lytic and latent. The latter is categorized into four programs (latency types 0–III) in which latent viral genes are expressed differentially depending on the tissue of origin and state of cells. The production of viral latent genes tends to decrease with an increase in time, and, in an approximate manner, the expression levels of viral genes are inversely correlated with the degree of abnormalities in the host genome (Fig. 1, Murata, Sato, Kimura, Rev Med Virol 2014). Execution of the viral lytic cycle contributes to not only progeny production, but also oncogenesis. EBV reactivation from latency is regulated by the balance between active and negative transcription factors. We recently identified, by functional screening, that MEF2, SP1, and bZip transcription factors play major roles in transcription of the EBV immediate early (IE) gene, BZLF1, and thereby in induction of lytic replication cycle (Fig. 2, Murata et al., J Virol 2013). Understanding the life cycle of the EBV and its relevance in oncogenesis may provide valuable clues to the development of effective therapies for the associated cancers.
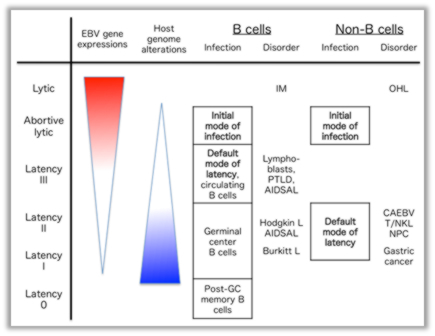
Fig. 1. Mode of infection and oncogenesis of the EBV.
The abortive lytic state is the initial mode of infection in B or non-B cells because of the pre-latent, transient lytic infection upon primary infection. Latency III and II are the default modes of latency in B and non-B cells, respectively. Although EBV gene expressions are silenced, slowly but steadily, the host genome accumulates genetic or epigenetic alterations for malignancies. IM, infectious mononucleosis; PTLD, post-transplant lymhoproliferative disorder; AIDSAL, AIDS-associated lymphomas; OHL, oral hairy leukoplakia; CAEBV, chronic active EBV infection; T/NKL, T/NK cell lymphomas, NPC, nasopharyngeal carcinoma
Fig. 2. Schematic representation of the minimal BZLF1 promoter.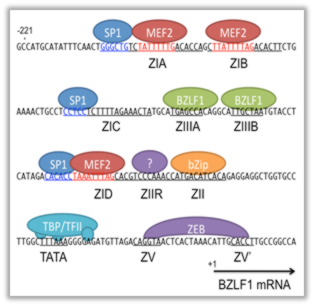
We have identified that MEF2, SP1, and bZip transcription factors can bind to and enhance BZLF1 promoter upon stimulation. Because BZLF1 is the crucial viral transcriptional activator of lytic cycle, activation of the BZLF1 promoter leads to reactivation of EBV from latency.Fi Fig. 2. Schematic representation of the minimal BZLF1 promoter. We have identified that MEF2, SP1, and bZip transcription factors can bind to and enhance BZLF1 promoter upon stimulation. Because BZLF1 is the crucial viral transcriptional activator of lytic cycle, activation of the BZLF1 promoter leads to reactivation of EBV from latency.g. 2. Schematic representation of the minimal BZLF1 promoter. We have identified that MEF2, SP1, and bZip transcription factors can bind to and enhance BZLF1 promoter upon stimulation. Because BZLF1 is the crucial viral transcriptional activator of lytic cycle, activation of the BZLF1 promoter leads to reactivation of EBV from latency.
RESEARCH PROJECTS
- Mechanism of how EBV can contribute to oncogenesis
- Development of anti-viral and anti-cancer drugs for EBV and EBV-associated cancers
- Functions of EBV genes
- Mechanism of transcriptional regulation of some viral genes that play key roles in EBV life cycle
- Application of HSV to oncolytic treatment of cancers
BIBLIOGRAPHY
2026
- Sato Y, Okuno Y, Murata T, Kimura H. Epstein–Barr virus-associated T/NK-cell neoplasms. J Med Virol, in press
- Kimura H, Cohen JI. Chronic Active Epstein–Barr Virus Disease: Molecular Pathogenesis, Evolving Concepts, and Emerging Therapies. Blood, in press
- Umeda M, Yoshimura K, Sato Y, Miyake Y, Kimura H Endosomal maturation leads to nucleocapsid conformation change in seasonal coronaviruses. Nagoya J Med Sci, in press
2025
- Bartlett A, Krejdovsky C, Yang G, Barry AP, Oduor C, Bailey JA, Moormann AM, Sato Y, Luftig MA. bioRxiv [Preprint]. 2025 Dec 19:2025.12.19.695354. doi: 10.64898/2025.12.19.695354. PMID: 41446165
- Sato Y, Okuno Y, Murata T, Kimura H. Defective But Tumorigenic: The Evolutionary and Functional Roles of Mutated Oncoviruses. FEMS Microbiol Rev. 2025 Jan 14:49:fuaf048. doi: 10.1093/femsre/fuaf048. PMID: 41004261
- Park H, Nakamura N, Miyamoto S, Sato Y, Kim KS, Kitagawa K, Kobashi Y, Tani Y, Shimazu Y, Zhao T, Nishikawa Y, Omata F, Kawashima M, Abe T, Saito Y, Nonaka S, Takita M, Yamamoto C, Morioka H, Kato K, Sagou K, Yagi T, Kawamura T, Sugiyama A, Nakayama A, Kaneko Y, Shibata RY, Aihara K, Kodama T, Kamiyama A, Tamura T, Fukuhara T, Shibuya K, Suzuki T, Iwami S, Tsubokura M. Longitudinal antibody titers measured after COVID-19 mRNA vaccination can identify individuals at risk for subsequent infection. Sci Transl Med. 2025 Sep 17;17(816):eadv4214. doi: 10.1126/scitranslmed.adv4214. Epub 2025 Sep 17. PMID: 40961221
- Aleksic B, Arthur A, Kasai-Prince M, Chretien B, Bustos-Villalobos I, Plewa C, Kasuya H, Page AJ, Zannettino ACW, Kimura H. The Nagoya University and the University of Adelaide Joint PhD Degree Program in Medicine - A Program of Opportunity. Adv Med Educ Pract. 2025 Sep 6;16:1635-1640. doi: 10.2147/AMEP.S526211. eCollection 2025.PMID: 40949886
- Kimura H, Chan JKC, Ng S-B, Sangueza JM, Tan SY. Severe mosquito bite allergy: WHO Classification of Tumours 5th ed., Vol.12, Skin Tumours. Who Classification of Tumours Editorial Board eds, WHO Press, Lyon, p430-431, 2025
- Kimura H, Chan JKC, Plaza JA, Sangueza JM. Hydroa vacciniforme lymphoproliferative disorder: WHO Classification of Tumours 5th ed., Vol.12, Skin Tumours. Who Classification of Tumours Editorial Board eds, WHO Press, Lyon, p432-433, 2025
- Khine HT, Sato Y, Hamada M, Umeda M, Iizuka A, Son S, Arai H, Kojima Y, Watanabe T, Naruse A, Goto K, Ohshima K, Akutsu Y, Nakaguro M, Satou A, Kataoka H, Ito Y, Sawada A, Kato S, Kawada JI, Murata T, Okuno Y, Kimura H. Association of Epstein–Barr viral genomic alterations to human pathologies. Blood. 2025 Sep 25;146(13):1533-1545. doi: 10.1182/blood.2024028055. PMID: 40569273
- Yabushita T, Yamashita D, Imai Y, Kimura H, Ishikawa T. Elderly-onset Hydroa Vacciniforme Lymphoproliferative Disorder. Int Med. 2025 Jul 24. doi: 10.2169/internalmedicine.5574-25. Online ahead of print. PMID: 40707220
- Sato Y, Hatanaka Y, Sato Y, Matsumoto K, Osana S, Nagatomi R, Nishizawa S. Viral Membrane-Targeting Amphipathic Helical Peptide-Based Fluorogenic Probes for the Analysis of Infectious Titers of Enveloped Viruses. Anal Chem. 2025 Mar 18;97(10):5480-5487. doi: 10.1021/acs.analchem.4c04852. Epub 2025 Jan 22. PMID: 39840494
- Kojima Y, Hamada M, Naruse A, Goto K, Khine HT, Arai H, Akutsu Y, Satou A, Nakaguro M, Kato S, Kodera Y, Yatabe Y, Torii Y, Kawada JI, Murata T, Kimura H, Takiguchi S, Inagaki H, Kataoka H, Okuno Y. The landscape of 142 Epstein-Barr viral whole genomes in gastric cancer. J Gastroenterol. 2025 Jan;60(1):55-65. doi: 10.1007/s00535-024-02170-3. Epub 2024 Nov 21. PMID: 39572460
2024
- Adachi Y, Terakura S, Osaki M, Okuno Y, Sato Y, Sagou K, Takeuchi Y, Yokota H, Imai K, Steinberger P, Leitner J, Hanajiri R, Murata M, Kiyoi H. Cullin-5 deficiency promotes chimeric antigen receptor T cell effector functions potentially via the modulation of JAK/STAT signaling pathway. Nat Commun. 2024 Dec 10;15(1):10376. doi: 10.1038/s41467-024-54794-x. PMID: 39658572
- Kimura H, Araujo I, Kato S, Ng S-B. Severe mosquito bite allergy: WHO Classification of Tumours Haematolymphoid tumours. Who Classification of Tumours Editorial Board eds. 5th ed, WHO Press, Lyon, p772-773, 2024
- Kimura H, Plaza JA, Araujo I, Rao HL, Chan JKC, Sangueza JM, Gru A. Hydroa vacciniforme lymphoproliferative disorder: WHO Classification of Tumours Haematolymphoid tumours. Who Classification of Tumours Editorial Board eds. 5th ed, WHO Press, Lyon, p774-776, 2024
- Araujo I, Kimura H, Liu WP, Sangueza OP. Systemic chronic active EBV disease: WHO Classification of Tumours Haematolymphoid tumours. Who Classification of Tumours Editorial Board eds. 5th ed, WHO Press, Lyon, p777-778, 2024
- Fujikawa Y, Kato K, Unno K, Narita S, Okuno Y, Sato Y, Takefuji M, Murohara T. Dynamic upregulation of retinoic acid signal in the early postnatal murine heart promotes cardiomyocyte cell cycle exit and maturation. Sci Rep. 2024 Aug 30;14(1):20222. doi: 10.1038/s41598-024-70918-1. PMID: 39215116
- Corrigendum: Inagaki T, Sato Y, Ito J, Takaki M, Okuno Y, Yaguchi M, Masud HMAA, Watanabe T, Sato K, Iwami S, Murata T, Kimura H. Direct evidence of abortive lytic infection-mediated establishment of Epstein-Barr virus latency during B-cell infection. Front Microbiol. 2024 May 23;15:1426311. doi: 10.3389/fmicb.2024.1426311. eCollection 2024. PMID: 38846570
- Nakamura N, Kobashi Y, Kim KS, Park H, Tani Y, Shimazu Y, Zhao T, Nishikawa Y, Omata F, Kawashima M, Yoshida M, Abe T, Saito Y, Senoo Y, Nonaka S, Takita M, Yamamoto C, Kawamura T, Sugiyama A, Nakayama A, Kaneko Y, Jeong YD, Tatematsu D, Akao M, Sato Y, Iwanami S, Fujita Y, Wakui M, Aihara K, Kodama T, Shibuya K, Iwami S, Tsubokura M. Modeling and predicting individual variation in COVID-19 vaccine-elicited antibody response in the general population. PLOS Digit Health. 2024 May 3;3(5):e0000497. doi: 10.1371/journal.pdig.0000497. eCollection 2024 May. PMID: 38701055
- Suzuki T, Sato Y, Okuno Y, Torii Y, Fukuda Y, Haruta K, Yamaguchi M, Kawamura Y, Hama A, Narita A, Muramatsu H, Yoshikawa T, Takahashi Y, Kimura H, Ito Y, Kawada JI. Single-cell transcriptomic analysis of Epstein-Barr virus-associated hemophagocytic lymphohistiocytosis. J Clin Immunol. 2024 Apr 20;44(4):103. doi: 10.1007/s10875-024-01701-0. PMID: 38642164
- Sagou K, Sato Y, Okuno Y, Watanabe T, Inagaki T, Motooka Y, Toyokuni S, Murata T, Kiyoi H, Kimura H. Epstein-Barr virus lytic gene BNRF1 promotes B-cell lymphomagenesis via IFI27 upregulation. PLoS Pathog. 2024 Feb 1;20(2):e1011954. doi: 10.1371/journal.ppat.1011954. eCollection 2024 Feb. PMID: 38300891
- Miyake Y, Hara Y, Umeda M, Banerjee I. Influenza A virus: Cellular entry. Subcell Biochem. 2023;106:387-401. doi: 10.1007/978-3-031-40086-5_14. PMID: 38159235
- Kimura H, Murata T. Understanding the Role of EBV Infection in Lymphomagenesis; Precision Cancer Therapies vol 2. O’Connor OA ed, Wiley-Blackwell, p235-45, 2024
2023
- Fukaura R, Terashima-Murase C, Tanahashi K, Sato Y, Kawase M, Kato K, Akiyama M. Generalized Verrucosis on a Background of 3C Syndrome Treated With Subcutaneous IgG Supplementation. JAMA Dermatol. 2023 Dec 1;159(12):1397-1399. doi: 10.1001/jamadermatol.2023.3801. PMID: 37851466
- Kawada JI, Ito Y, Ohshima K, Yamada M, Kataoka S, Muramatsu H, awada A, Wada T, Imadome KI, Arai A, Iwatsuki K, Ohga S, Kimura H. Updated guidelines for chronic active Epstein-Barr virus disease. Int J Hematol. 2023 Nov;118(5):568-576. doi: 10.1007/s12185-023-03660-5. Epub 2023 Sep 20. PMID: 37728704
- Taga S, Suga H, Nakano T, Kuwahara A, Inoshita N, Kodani Y, Nagasaki H, Sato Y, Tsumura Y, Sakakibara M, Soen M, Miwata T, Ozaki H, Kano M, Watari K, Ikeda A, Yamanaka M, Takahashi Y, Kitamoto S, Kawaguchi Y, Miyata T, Kobayashi T, Sugiyama M, Onoue T, Yasuda Y, Hagiwara D, Iwama S, Tomigahara Y, Kimura T, Arima H. Generation and purification of ACTH-secreting hPSC-derived pituitary cells for effective transplantation. Stem Cell Reports. 2023 Aug 8;18(8):1657-1671. doi: 10.1016/j.stemcr.2023.05.002. Epub 2023 Jun 8. PMID: 37295423
- Sugimoto A, Watanabe T, Matsuoka K, Okuno Y, Yanagi Y, Narita Y, Mabuchi S, Nobusue H, Sugihara E, Hirayama M, Ide T, Onouchi T, Sato Y, Kanda T, Saya H, Iwatani Y, Kimura H, Murata T. Growth Transformation of B cells by Epstein-Barr Virus Requires IMPDH2 Induction and Nucleolar Hypertrophy. Microbiol Spectr. 2023 Aug 17;11(4):e0044023. doi: 10.1128/spectrum.00440-23. Epub 2023 Jul 6. PMID: 37409959
- Uddin MK, Watanabe T, Arata M, Sato Y, Kimura H, Murata T. Epstein-Barr virus BBLF1 mediates secretory vesicle transport to facilitate mature virion release. J Virol. 2023 May 17:e0043723. doi: 10.1128/jvi.00437-23. Epub 2023 May 17. PMID: 37195206
- Hirai Y, Asada H, Hamada T, Kawada JI, Kimura H, Arai A, Ohshima K, Ohga S, Iwatsuki K. Diagnostic and disease severity determination criteria for hydroa vacciniforme lymphoproliferative disorders and severe mosquito bite allergy. J Dermatol. 2023 Jul;50(7):e198-e205. doi: 10.1111/1346-8138.16842. Epub 2023 May 30. PMID: 37249004.
- Sawa M, Murase C, Yamada N, Fukaura R, Tetsuka N, Sato Y, Takeichi T, Akiyama M. Giant condyloma of Buschke-Löwenstein in a Netherton syndrome patient, successfully treated with cryotherapy and intravenous immunoglobulin. J Dermatol. 2023 Apr 5. doi: 10.1111/1346-8138.16798. Online ahead of print. PMID: 37017444
- Nakaya Y, Koh H, Harada N, Okamura H, Misugi T, Suekane T, Tachibana D, Kimura H, Hino M, Nakamae H. Salvage HLA-haploidentical hematopoietic cell transplantation with post-transplant cyclophosphamide for graft failure in a patient with chronic active Epstein-Barr virus infection. Exp Clin Transplant. 2023 Feb;21(2):184-188. doi: 10.6002/ect.2022.0322. PMID: 36919727
- Murata T, Iwahori S, Okuno Y, Nishitsuji H, Yanagi Y, Watashi K, Wakita T, Kimura H, Shimotohno K. N6-methyladenosine modification of hepatitis B virus RNA in the coding region of HBx. Int J Mol Sci. 2023 Jan 23;24(3):2265. doi: 10.3390/ijms24032265. PMID: 36768585
- Kimura H. The 100th anniversary of the Nagoya Journal of Medical Science: a message from the Dean. Nagoya J. Med. Sci. 85. 5–6, 2023 doi:10.18999/nagjms.85.1.5. PMID: 36923615
2022
- Ding W, Wang C, Narita Y, Wang H, Leong MML, Huang A, Liao Y, Liu X, Okuno Y, Kimura H, Gewurz B, Teng M, Jin S, Sato Y, Zhao B. The Epstein-Barr Virus Enhancer Interaction Landscapes in Virus-Associated Cancer Cell Lines. J Virol. 2022 Sep 12:e0073922. doi: 10.1128/jvi.00739-22. Online ahead of print. PMID: 36094314
- Hara Y, Watanabe T, Yoshida M, Masud HMAA, Kato H, Kondo T, Suzuki R, Kurose S, Uddin MK, Arata M, Miyagi S, Yanagi Y, Sato Y, Kimura H, Murata T. Comprehensive Analyses of Intraviral Epstein-Barr Virus Protein-Protein Interactions Hint Central Role of BLRF2 in the Tegument Network. J Virol. 2022 Jul 27;96(14):e0051822. doi: 10.1128/jvi.00518-22. Epub 2022 Jul 11. PMID: 35862711
- Kimura H, de Leval L, Cai Q, Kim WS. EBV-associated NK and T-cell lymphoid neoplasms. Curr Opin Oncol. 2022 Sep 1;34(5):422-431. doi: 10.1097/CCO.0000000000000889. Epub 2022 Jul 27. PMID: 35900729
- Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IBO, Berti E, Bhagat G, Borges AM, Boyer D, Calaminici M, Chadburn A, Chan JKC, Cheuk W, Chng WJ, Choi JK, Chuang SS, Coupland SE, Czader M, Dave SS, )de Jong D, Du MQ, Elenitoba-Johnson KS, Ferry J, Geyer J, Gratzinger D, Guitart J, Gujral S, Harris M, Harrison CJ, Hartmann S, Hochhaus A, Jansen PM, Karube K, Kempf W, Khoury J, Kimura H, Klapper W, Kovach AE, Kumar S, Lazar AJ, Lazzi S, Leoncini L, Leung N, Leventaki V, Li XQ, Lim MS, Liu WP, Louissaint A Jr, Marcogliese A, Medeiros LJ, Michal M, Miranda RN, Mitteldorf C, Montes-Moreno S, Morice W, Nardi V, Naresh KN, Natkunam Y, Ng SB, Oschlies I, Ott G, Parrens M, Pulitzer M, Rajkumar SV, Rawstron AC, Rech K, Rosenwald A, Said J, Sarkozy C, Sayed S, Saygin C, Schuh A, Sewell W, Siebert R, Sohani AR, Tooze R, Traverse-Glehen A, Vega F, Vergier B, Wechalekar AD, Wood B, Xerri L, Xiao W. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia. 2022 Jul;36(7):1720-1748. doi: 10.1038/s41375-022-01620-2. Epub 2022 Jun 22. PMID: 35732829
- Suzuki T, Sato Y, Okuno Y, Goshima F, Mikami T, Umeda M, Murata T, Miyake Y, Watanabe T, Watashi K, Wakita T, Kitagawa H, Kimura H. Genome-wide CRISPR screen for HSV-1 host factors reveals PAPSS1 contributes to heparan sulfate synthesis. Commun Biol. 2022 Jul 19;5(1):694. doi: 10.1038/s42003-022-03581-9. PMID: 35854076 プレスリリース/解説
- Narita S, Unno K, Kato K, Okuno Y, Sato Y, Tsumura Y, Fujikawa Y, Shimizu Y, Hayashida R, Kondo K, Shibata R, Murohara T. Direct reprogramming of adult adipose-derived regenerative cells toward cardiomyocytes using six transcriptional factors. iScience. 2022 Jun 24;25(7):104651. doi: 10.1016/j.isci.2022.104651. eCollection 2022 Jul 15. PMID: 35811849
- Sato Y, Yaguchi M, Okuno Y, Ishimaru H, Sagou K, Ozaki S, Suzuki T, Inagaki T, Umeda M, Watanabe T, Fujimuro M, Murata T, Kimura H. Epstein-Barr virus tegument protein BGLF2 in exosomes released from virus-producing cells facilitates de novo infection. Cell Commun Signal. 2022 Jun 21;20(1):95. doi: 10.1186/s12964-022-00902-7. PMID: 35729616
- Yanagi Y, Watanabe T, Hara Y, Sato Y, Miyake Y, Kimura H, Murata T. EBV exploits RNA m6A modification for cell survival and efficient progeny virus production in lytic cycle. Front Microbiol. 2022 Jun 15;13:870816. doi: 10.3389/fmicb.2022.870816. eCollection 2022. PMID: 35783391
- Nishikubo M, Hiramoto N, Yamashita D, Imoto H, Sato Y, Okuno Y, Haga H, Nannya Y, Ogawa S, Kimura H, Ishikawa T. Elderly-onset systemic Epstein Barr virus-positive T cell lymphoma of childhood: a case report. Pathol Int. 2022 Jul;72(7):376-378. doi: 10.1111/pin.13231. Epub 2022 May 24. PMID: 35611496.
- Kondo S, Okuno Y, Murata T, Dochi H, Wakisaka N, Mizokami H, Moriyama-Kita M, Kobayashi E, Kano, M Komori T, Hirai N, Ueno T, Nakanishi Y, Endo K, Sugimoto H, Kimura H, Yoshizaki T. EBV genome variations enhance clinicopathological features of nasopharyngeal carcinoma in a non-endemic region. Cancer Sci. 2022 Jul;113(7):2446-2456. doi: 10.1111/cas.15381. Epub 2022 May 24. PMID: 35485636
- Wang L, Moreira EA, Kempf G, Miyake Y, Oliveira Esteves BI, Fahmi A, Schaefer JV, Dreier B, Yamauchi Y, Alves MP, Plückthun A, Matthias P. Disrupting the HDAC6-ubiquitin interaction impairs infection by influenza and Zika virus and cellular stress pathways. Cell Rep. 2022 Apr 26;39(4):110736. doi: 10.1016/j.celrep.2022.110736. PMID: 35476995
- Yanagi Y, Hara Y, Mabuchi S, Watanabe T, Sato Y, Kimura H, Murata T. PD-L1 Upregulation by Lytic Induction of Epstein-Barr virus. Virology 2022 2022 Mar;568:31-40. doi: 10.1016/j.virol.2022.01.006. PMID: 35093708
- Fujimoto KJ, Hobbs DCF, Umeda M, Nagata A, Yamaguchi R, Sato Y, Sato S, Ohmatsu K, Ooi T, Yanai T, Kimura H, Murata T. In silico Analysis and Synthesis of Nafamostat Derivatives and Evaluation of Their Anti-SARS-CoV-2 Activity. Viruses 2022 Feb 14;14(2):389. doi: 10.3390/v14020389. PMID: 35215982
2021
- Murata T, Sugimoto A, Inagaki T, Watanabe T, Sato Y, Kimura H. Molecular Basis of Epstein-Barr Virus Latency Establishment and Lytic Reactivation. Viruses 2021 Nov 23;13(12):2344. doi: 10.3390/v13122344. PMID: 34960613
- Miyagi S, Watanabe T, Hara Y, Arata M, Uddin MMK, Mantoku K, Sago K, Yusuke Yanagi Y, Suzuki T, Masud HMAA , Kawada JI, Nakamura S, Miyake Y, Sato Y, Murata T, Kimura H. A STING inhibitor suppresses EBV-induced B cell transformation and lymphomagenesis. Cancer Sci 2021 Oct 5. doi: 10.1111/cas.15152. Online ahead of print. PMID: 34609775.
- Oka K, Morioka H, Eguchi M, Sato Y, Tetsuka N, Iguchi M, Kanematsu T, Fukano H, Hoshino Y, Kiyoi H, Yagi T. Bursitis, Bacteremia, and Disseminated Infection of Mycobacteroides (Mycobacterium) abscessus subsp. massiliense. Intern Med. 2021 Sep 15;60(18):3041-3045. doi: 10.2169/internalmedicine.6189-20. Epub 2021 Mar 29. PMID: 33775997
- Kimura H, Okuno Y, Sato Y, Watanabe T, Murata T. Deletion of Viral microRNAs in the Oncogenesis of Epstein-Barr Virus-Associated Lymphoma. Front Microbiol. 2021 Jul 8;12:667968. doi: 10.3389/fmicb.2021.667968. eCollection 2021. PMID: 34305835
- Yanagi Y, Okuno Y, Narita Y, Masud HMA, Watanabe T, Sato Y, Kanda T, Kimura H, Murata T. RNAseq analysis identifies involvement of EBNA2 in PD-L1 induction during Epstein-Barr virus infection of primary B cells. Virology. 2021 Feb 21;557:44-54. doi: 10.1016/j.virol.2021.02.004. Online ahead of print. PMID: 33639481
- Mabuchi S, Hijioka F, Watanabe T, Yanagi Y, Okuno Y, Masud HMA, Sato Y, Murata T, Kimura H. Role of Epstein-Barr virus C promoter deletion found in diffuse large B cell lymphoma. Cancers (Basel). 2021 Feb 1;13(3):561. doi: 10.3390/cancers13030561. PMID: 33535665
- Inagaki T, Sato Y, Ito, Takaki M, Okuno Y, Yaguchi M, Masud HMA, Watanabe T, Sato K, Iwami S, Murata T, Kimura H. Direct evidence of abortive lytic infection-mediated establishment of Epstein-Barr virus latency during B-cell infection. Front Microbiol., 2021 Jan 21;11:575255. doi: 10.3389/fmicb.2020.575255. eCollection 2020. PMID: 33613459
2020
- Takano G, Esaki S, Goshima F, Enomoto A, Hatano Y, Ozaki H, Watanabe T, Sato Y, Kawakita D, Murakami S, Murata T, Nishiyama Y, Iwasaki S, Kimura H. Oncolytic activity of naturally attenuated herpes-simplex virus HF10 against an immunocompetent model of oral carcinoma. Molecular Therapy – Oncolytics, 2020 Dec 19;20:220-227. doi: 10.1016/j.omto.2020.12.007. eCollection 2021 Mar 26. PMID: 33665360
- Sato Y, Iguchi M, Kato Y, Morioka H, Hirabayashi A, Tetsuka N, Tomita Y, Kato D, Yamada K, Kimura H, Yagi T. Number of concomitant drugs with thrombocytopenic adverse effect and the extent of resolution of inflammatory response are risk factors for thrombocytopenia in patients treated with Linezolid for more than 14 days. Nagoya J Med Sci 2020 Aug;82(3):407-414. doi: 10.18999/nagjms.82.3.407. PMID: 33132425
- Esaki E, Goshima F, Ozaki H, Takano G, Hatano Y, Kawakita D, Ijichi K, Watanabe T, Sato Y, Murata T, Iwata H, Shibamoto Y, Murakami S, Nishiyama Y, Kimura H. Oncolytic activity of HF10 in head and neck squamous cell carcinomas. Cancer Gene Ther 2020 Aug;27(7-8):585-598. doi: 10.1038/s41417-019-0129-3. PMID: 31477804
- Yonese I, Sakashita C, Imadome KI, Kobayashi T, Yamamoto M, Sawada A, Ito Y, Fukuhara N, Hirose A, Takeda Y, Makita M, Endo T, Kimura SI, Ishimura M, Miura O, Ohga S, Kimura H, Fujiwara S, Arai A. Nationwide survey of systemic chronic active EBV infection in Japan in accordance with the new WHO classification. Blood Adv. 2020 Jul 14;4(13):2918-2926. doi: 10.1182/bloodadvances.2020001451. PMID: 32598475
- Shibata A, Ishiguro Y, Makita S, Yamaga Y, Kimura H, Akiyama M. A systemic form chronic active Epstein-Barr virus infection diagnosed from erythema nodosum-like skin lesions. Eur J Dermatol. 2020 Jun 1;30(3):314-316. doi: 10.1684/ejd.2020.3781. PMID: 32666933
- Murata T, Okuno Y, Sato Y, Watanabe T, Kimura H. Oncogenesis of CAEBV Revealed: Intragenic Deletions in the Viral Genome and Leaky Expression of Lytic Genes\\\". Rev Med Virol. 2020 Mar;30(2):e2095. doi: 10.1002/rmv.2095. PMID: 31845495
- Cohen JI, Iwatsuki K, Ko YH, Kimura H, Manoli I, Ohshima K, Pittaluga S, Quintanilla-Fend L, Jaffe ES. Epstein-Barr virus NK and T cell lymphoproliferative disease: report of a 2018 international meeting. Lymphoma Leuk 2020 Apr;61(4):808-819. doi: 10.1080/10428194.2019.1699080. PMID: 31833428
- Watanabe T, Sato Y, Masud HMA, Takayama M, Matsuda H, Hara Y, Yanagi Y, Yoshida M, Goshima F, Murata T, Kimura H. Antitumor activity of CDK inhibitor alsterpaullone in Epstein-Barr virus-associated lymphoproliferative disorders. Cancer Sci 2020 Jan;111(1):279-287. doi: 10.1111/cas.14241. PMID: 31743514
2019
- Iemura T, Kondo T, Hishizawa M, Yamashita K, Kimura H, Takaori-Kondo A. NK-cell post-transplant lymphoproliferative disease with chronic active Epstein-Barr virus infection-like clinical findings. Int J Infect Dis. 2019 Nov;88:31-33. doi: 10.1016/j.ijid.2019.07.039. Epub 2019 Aug 6. PMID: 31398454
- Fujiwara S, Kimura H. Editorial: Epstein-Barr virus-associated T/NK-cell lymphoproliferative diseases. Frontiers Pediatr. 2019 Jul 10;7:285. doi: 10.3389/fped.2019.00285. PMID: 31355168
- Kawada JI, Kamiya Y, Sawada A, Iwatsuki K, Izutsu K, Torii Y, Kimura H, Ito Y. Viral DNA loads in various blood components of patients with Epstein–Barr virus-positive-T/NK cell lymphoproliferative diseases. J Infect Dis. 2019 Sep 13;220(8):1307-1311. doi: 10.1093/infdis/jiz315. PMID: 31240305
- Masud HMA, Watanabe T, Sato Y, Goshima F, Kimura H, Murata T. The BOLF1 Gene is Necessary for Effective Epstein-Barr Viral Infectivity. Virology. 2019 May;531:114-125. doi: 10.1016/j.virol.2019.02.015. PMID: 30856483
- Miyake Y, Keusch JJ, Decamps L, Ho-Xuan H, Iketani S, Gut H, Kutay U, Helenius A, Yamauchi Y. Influenza virus uses transportin 1 for vRNP debundling during cell entry. Nat Microbiol. 2019 Apr;4(4):578-586. doi: 10.1038/s41564-018-0332-2 PMID: 30692667
- Sato Y, Watanabe T, Suzuki C, Abe Y, Masud HMA, Inagaki T, Yoshida M, Suzuki T, Goshima F, Adachi J, Tomonaga T, Murata T, Kimura H. S-like phase CDKs stabilize the Epstein-Barr virus BDLF4 protein to temporally control late gene transcription. J Virol. 2019 Apr 3;93(8). pii: e01707-18. doi: 10.1128/JVI.01707-18. PMID: 30700607
- Yanagi Y, Masud HMA, Watanabe T, Sato Y, Goshima F, Kimura H, Murata T. Initial Characterization of the Epstein-Barr Virus BSRF1 Gene Product. Viruses. 2019 Mar 21;11(3). pii: E285. doi: 10.3390/v11030285. PMID: 30901892
- Okuno Y, Murata Y, Sato Y, Muramatsu H, YIto Y, Watanabe T, Okuno T, Murakami N, Yoshida K, Sawada A, Inoue M, Kawa K, Seto M, Ohshima K, Shiraishi Y, Chiba K, Tanaka H, Miyano S, Narita Y, Yoshida M, Goshima F, Kawada JI, Nishida T, Kiyoi H, Kato S, Nakamura S, Morishima S, Yoshikawa T, Fujiwara S, Shimizu N, Isobe Y, Noguchi M, Kikuta1 A, Iwatsuki K, Takahashi Y, Kojima S, Ogawa S, Kimura H. Defective Epstein-Barr virus (EBV) in chronic active EBV infection and EBV-related hematological malignancy. Nat Microbiol. 2019 Mar;4(3):404-413. doi: 10.1038/s41564-018-0334-0. PMID: 30664667
- Kimura H, Kwong YL. EBV Viral Loads in Diagnosis, Monitoring, and Response Assessment. Frontiers Oncol. 2019 Feb 12;9:62. doi: 10.3389/fonc.2019.00062. PMID: 30809508
- Kimura H, Fujiwara S. Overview of EBV-associated T/NK-cell lymphoproliferative diseases. Frontiers Pediatr. 2019 Jan 4;6:417. doi: 10.3389/fped.2018.00417. PMID: 30662890
2018
- Ichikawa T, Okuno Y, Sato Y, Goshima F, Yoshiyama H, Kanda T, Kimura H, Murata T. Regulation of EBV Lifecycle and Cell Proliferation by Histone H3K27 Methyltransferase, EZH2, in Akata Cells. mSphere. 2018 Nov 28;3(6). pii: e00478-18. doi: 10.1128/mSphere.00478-18. PMID: 3048715
- Kimura H. JAK inhibitors for refractory lymphoma. Oncotarget 2018, Vol. 9, (No. 68), pp: 32883-32884 doi: 10.18632/oncotarget.26054
- Murata T. Encyclopedia of EBV-Encoded Lytic Genes: An Update. Adv Exp Med Biol. 2018;1045:395-412. doi: 10.1007/978-981-10-7230-7_18. PMID: 29896677
- Watanabe D, Goshima F. Oncolytic Virotherapy by HSV. Adv Exp Med Biol. 2018;1045:63-84. doi: 10.1007/978-981-10-7230-7_4. PMID: 29896663
- Kimura H. EBV in T-/NK-Cell Tumorigenesis. Adv Exp Med Biol. 2018;1045:459-475. doi: 10.1007/978-981-10-7230-7_21. PMID: 29896680
- Konishi N, Narita Y, Hijioka F, Masud HMAA, Sato Y, Kimura H, Murata M. BGLF2 Increases Infectivity of Epstein-Barr virus by Activating AP-1 upon de novo Infection. mSphere 2018 Apr 25;3(2). pii: e00138-18. doi: 10.1128/mSphere.00138-18. PMID: 29695622
- Miyake Y, Matthias P, Yamauchi Y. Purification of Unanchored Polyubiquitin Chains from Influenza Virions. Methods Mol Biol. 2018;1836:329-342. doi: 10.1007/978-1-4939-8678-1_16. PMID: 30151581
- Matsuzawa T, Nakamura Y, Ogawa Y, Ishimaru K, Goshima F, Shimada S, Nakao A, Kawamura T. Differential Day-Night Outcome to HSV-2 Cutaneous Infection. J Invest Dermatol. 2018 Jan;138(1):233-236. doi: 10.1016/j.jid.2017.07.838. PMID: 28842321
- Kawamoto K, Miyoshi H, Suzuki T, Kozai Y, Kato K, Miyahara M, Yujiri T, Oishi N, Choi I, Fujimaki K, Muta T, Kume M, Moriguchi S, Tamura S, Kato T, Tagawa H, Makiyama J, Kanisawa Y, Sasaki Y, Kurita D, Yamada K, Shimono J, Sone H, Takizawa J, Seto M, Kimura H, Ohshima K. A distinct subtype of Epstein Barr virus positive T/NK-cell lymphoproliferative disorder: Adult patients with chronic active Epstein Barr virus infection-like features. Haematologica. 2018 Jun;103(6):1018-1028. doi: 10.3324/haematol.2017.174177. PMID: 29242302
- Kawada JI, Ando S, Torii Y, Ito Y, Kimura H. Antitumor effects of duvelisib on Epstein-Barr virus-associated lymphoma cells. Cancer Med 2018 Apr;7(4):1275-1284. doi: 10.1002/cam4. PMID: 29522278
2017
- Kimura H, Cohen JI. Chronic Active Epstein-Barr Virus Disease. Front Immunol 8:1867, 2017 doi: 10.3389/fimmu.2017.01867
- Yoshida M, Murata T, Ashio K, Narita Y, Watanabe T, Masud HMAA, Sato Y, Goshima F, Kimura H. Characterization of a Suppressive Cis-acting Element in the Epstein-Barr virus LMP1 Promoter. Front Microbiol 8: 2302, 2017
- Masud HMA, Watanabe T, Yoshida M, Sato Y, Goshima F, Kimura H, Murata T. Epstein-Barr Virus BKRF4 Gene Product Is Required for Efficient Progeny Production. J Virol doi: 10.1128/JVI.00975-17. [Epub ahead of print]
- Quintanilla-Martinez L, Ko YH, Kimura H, Jaffe ES. EBV-positive T-cell and NK-cell lymphoproliferative diseases of childhood: WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J eds. Revised 4th ed, IARC Press, Lyon, p355-62, 2017
- Tanaka R, Goshima F, Esaki S, Sato Y, Murata T, Nishiyama Y, Watanabe D, Kimura H. The efficacy of combination therapy with oncolytic herpes simplex virus HF10 and dacarbazine in a mouse melanoma model. Am J Cancer Res 7:1693-1703, 2017
- Yoshida M, Watanabe T, Narita Y, Sato Y, Goshima F, Kimura H, Murata T. The Epstein-Barr Virus BRRF1 Gene is Dispensable for Viral Replication in HEK293 cells and Transformation. Sci Rep 7:6044, 2017
- Sato Y, Ochiai S, Murata T, Kanda T, Goshima F, Kimura H. Elimination of LMP1-expressing cells from a monolayer of gastric cancer AGS cells. Oncotarget 8:39345-39355, 2017
- Washio K, Oka T, Abdalkader L, Muraoka M, Shimada A, Oda M, Sato H, Takata K, Kagami Y, Shimizu N, Seto M, Nakamura S, Kimura H, Yoshino T, Tsukahara H. Gene expression analysis of hypersensitivity to mosquito bite, chronic active EBV infection and NK/T-lymphoma/leukemia. Leuk Lymphoma 58: 2683-2694, 2017
- Watanabe T, Sakaida K, Yoshida M, Masud HMA, Sato Y, Goshima F, Kimura H, Murata T. The C-terminus of Epstein-Barr virus BRRF2 is required for its proper localization and efficient virus production. Front Microbiol 8: 125, 2017
- Torii Y, Kawada JI, Murata T, Yoshiyama H, Kimura H, Ito Y. Epstein-Barr virus infection-induced inflammasome activation in human monocytes. PLoS One 12(4):e0175053, 2017
2016
- Murata T, Noda C, Narita Y, Watanabe T, Yoshida M, Ashio K, Sato Y, Goshima F, Kanda T, Yoshiyama H, Tsurumi T, Kimura H. Induction of Epstein-Barr Virus Oncoprotein Latent Membrane Protein 1 (LMP1) by Transcription Factors Activating Protein 2 (AP-2) and Early B Cell Factor (EBF). J Virol 90:3873-89, 2016
- Ando S, Kawada JI, Watanabe T, Suzuki M, Sato Y, Torii Y, Asai M, Goshima F, Murata T, Shimizu N, Ito Y, Kimura H. Tofacitinib induces G1 cell-cycle arrest and inhibits tumor growth in Epstein-Barr virus-associated T and natural killer cell lymphoma cells. Oncotarget 7:76793-76805, 2016
- Kawano Y, Kawada JI, Kamiya Y, Suzuki M, Torii Y, Kimura H, Ito Y. Analysis of circulating human and viral microRNAs in patients with congenital cytomegalovirus infection. J Perinatol 36: 1101-1105, 2016
- Aoki R, Kawamura T, Goshima F, Ogawa Y, Nakae S, Moriishi K, Nakao A, Shimada S. The Alarmin IL-33 Derived from HSV-2-Infected Keratinocytes Triggers Mast Cell-Mediated Antiviral Innate Immunity. J Invest Dermatol 136(6):1290-2, 2016.
- Kawamura Y, Miura H, Matsumoto Y, Uchida H, Kudo K, Hata T, Ito Y, Kimura H, Yoshikawa T. A case of Epstein-Barr virus–associated hemophagocytic lymphohistiocytosis with severe cardiac complications. BMC Pediatrics 16:172, 2016
- Ikumi K, Ando T, Katano H, Katsuno M, Sakai Y, Yoshida M, Saida T, Kimura H, Sobue G. HSV-2-related hemophagocytic lymphohistiocytosis in a fingolimod-treated MS patient. Neurol Neuroimmunol Neuroinflamm 3(4):e247, 2016
- Shimomura M, Morishita H, Meguro T, Seto S, Kimura M, Hamazaki M, Hashimoto A, Sugiyama Y, Kimura H. A case of CAEBV showing features of granulomatosis with polyangiitis. Pediatr Int 58:639-42, 2016
- Ito Y, Suzuki M, Kawada J, Kimura H. Diagnostic values for the viral load in peripheral blood mononuclear cells of patients with chronic active Epstein-Barr virus disease. J Infect Chemother 22:268-271, 2016
2015
- Watanabe T, Narita Y, Yoshida M, Sato Y, Goshima F, Kimura H, Murata T. Epstein-Barr virus BDLF4 gene is required for efficient expression of viral late lytic genes. J Virol 89(19):10120-4
- Narita Y, Sugimoto A, Kawashima D, Watanabe T, Kanda T, Kimura H, Tsurumi T, Murata T. A herpesvirus specific motif of Epstein-Barr virus DNA polymerase is required for the efficient lytic genome synthesis. Sci Rep 5:11767, 2015
- Watanabe T, Tsuruoka M, Narita Y, Katsuya R, Goshima F, Kimura H, Murata T. The Epstein-Barr virus BRRF2 gene product is involved in viral progeny production. Virology 484:33-40, 2015
- Watanabe T, Fuse K, Takano T, Narita Y, Goshima F, Kimura H, Murata T. Roles of Epstein-Barr virus BGLF3.5 Gene and Two Upstream Open Reading Frames in Lytic Viral Replication in HEK293 Cells. Virology 483: 44–53, 2015
- Suzuki M, Takeda T, Nakagawa H, Iwata S, Watanabe T, Siddiquey MN, Goshima F, Murata T, Kawada JI, Ito Y, Kojima S, Kimura H. The heat shock protein 90 inhibitor BIIB021 suppresses the growth of T and natural killer cell lymphomas.Front Microbiol 6:280, 2015
- Nagata K, Nakayama Y, Higaki K, Ochi M, Kanai K, Matsushita M, Kuwamoto S, Kato M, Murakami I, Iwasaki T, Nanba E, Kimura H, Hayashi K. Reactivation of persistent Epstein—Barr virus (EBV) causes secretion of thyrotropin receptor antibodies (TRAbs) in EBVinfected B lymphocytes with TRAbs on their surface. Autoimmunity 11:1-8, 2015
- Morishima S, Nakamura S, Yamamoto K, Miyauchi H, Kagami Y, Kinoshita T, Onoda H, Yatabe Y, Ito M, Miyamura K, Nagai H, Moritani S, Sugiura I, Tsushita K, Mihara H, Ohbayashi K, Iba S, Emi N, Okamoto M, Iwata S, Kimura H, Kuzushima K, Morishima Y. Increased T-cell responses to EBV with high viral load in patients with EBV-positive diffuse large B-cell lymphoma.Leukemia & Lymphoma 56:1072-8, 2015
- Coleman CB, Wohlford EM, Smith NA, King CA, Ritchie JA, Baresel PC, Kimura H, Rochford R. Epstein-Barr virus Type 2 latently infects T-cells inducing an atypical activation characterized by expression of lymphotactic cytokines. J Virol 89:2301-12, 2015
- Kawada J, Torii Y, Kawano Y, Suzuki M, Kamiya Y, Kotani T, Kikkawa F, Kimura H, Ito Y. Viral load in children with congenital cytomegalovirus infection identified on newborn hearing screening. J Clin Virol 65:41-5, 2015
- Kawano Y, Suzuki M, Kawada JI, Kimura H, Kamei H, Ohnishi Y, Ono Y, Uchida H, Ogura Y, Ito Y. Effectiveness and safety of immunization with live-attenuated and inactivated vaccines for pediatric liver transplantation recipients. Vaccine 33:1440-5, 2015
2014
- Kanazawa T, Hiramatsu Y, Iwata S, Siddiquey MN, Sato Y, Suzuki M, Ito Y, Goshima F, Murata T, Kimura H. Anti-CCR4 monoclonal antibody mogamulizumab for the treatment of EBV-associated T- and NK-cell lymphoproliferative diseases. Clin Cancer Res 20:5075-84, 2014
- Kawada JI, Ito Y, Iwata S, Suzuki M, Kawano Y, Kanazawa T, Siddiquey MN, Kimura H. mTOR inhibitors induce cell cycle arrest and inhibit tumor growth in Epstein-Barr virus-associated T and natural killer cell lymphoma cells. Clin Cancer Res 20:5412-22, 2014
- Murata T, Sato Y, Kimura H. Modes of infection and oncogenesis by the Epstein-Barr virus. Rev Med Virol 24: 242–253, 2014
- Murata T. Regulation of Epstein-Barr virus reactivation from latency. Microbiol Immunol 58(6):307-17, 2014
- Fujiwara S, Kimura H, Imadome KI, Arai A, Kodama E, Morio T, Shimizu N, Wakiguchi H. Current Studies on Chronic Active Epstein-Barr virus Infection in Japan. Pediatr Int 56, 159–166, 2014
- Ito T, Kawazu H, Murata T, Iwata S, Arakawa S, Sato Y, Kuzushima K, Goshima F, Kimura H. Role of latent membrane protein 1 (LMP1) in chronic active Epstein-Barr virus infection (CAEBV)-derived T/NK cell proliferation. Cancer Med 3: 787-795, 2014
- Siddiquey MN, Nakagawa H, Iwata S, Kanazawa T, Suzuki M, Imadome KI, Fujiwara S, Goshima F, Murata T, Kimura H. Anti-tumor effects of suberoylanilide hydroxamic acid on Epstein-Barr virus-associated T- and natural killer- cell lymphoma. Cancer Sci 105:713-722, 2014
- Murata T, Tsurumi T. Switching of EBV cycles between latent and lytic states. Rev Med Virol 24:142-53, 2014
- Kimura H, Karube K, Ito Y, Hirano K, Suzuki M, Iwata S, Seto M. Rare occurrence of JAK3 mutations in NK cell neoplasms in Japan. Leukemia & Lymphoma 55:962-3, 2014
- Goshima F, Esaki S, Luo C, Kamakura M, Kimura H, Nishiyama Y. Oncolytic viral therapy with a combination of HF10, a herpes simplex virus type 1 variant, and granulocyte–macrophage colony-stimulating factor for murine ovarian cancer. Int J Cancer 134:2865-77, 2014
- Nagata K, Higaki K, Nakayama Y, Miyauchi H, Kiritani Y, Kanai K, Matsushita M, Iwasaki T, Sugihara H, Kuwamoto S, Kato M, Murakami I, Nanba E, Kimura H, Hayashi K. Presence of Epstein-Barr virus-infected B lymphocytes with thyrotropin receptor antibodies on their surface in Graves' disease patients and in healthy individuals. Autoimmunity 47:193-200, 2014
2013
- Goshima F, Esaki S, Luo C, Kamakura M, Kimura H, Nishiyama Y.Oncolytic viral therapy with a combination of HF10, a herpes simplex virus type 1 variant, and granulocyte–macrophage colony-stimulating factor for murine ovarian cancer. Int J Cancer in press
- Kimura H, Karube K, Ito Y, Hirano K, Suzuki M, Iwata S, Seto M. Rare occurrence of JAK3 mutations in NK cell neoplasms in Japan. Leukemia & Lymphoma in press
- Kato S, Miyata T, Takata K, Shimada S, Ito Y, Tomita A, Elsayed AA, Takahashi E, Asano N, Kinoshita T, Kimura H, Nakamura S. Epstein-Barr virus-positive cytotoxic T-cell lymphoma followed by chronic active Epstein-Barr virus infection-associated T/NK-cell lymphoproliferative disorder: a case report. Hum Pathol in press
- Hayashi K, Ishigami M, Ishizu Y, Kuzuya T, Honda T, Itoh A, Hirooka Y, Ishikawa T, Nakano I, Ito Y, Kimura H, Katano Y, Goto H. A pediatric case of hepatitis B virus subgenotype A2 in Japan. Clin J Gastroenterol in press
- Kimura H, Kawda J, Ito Y. Epstein-Barr virus-associated lymphoid malignancies: the expanding spectrum of hematopoietic neoplasms. Nagoya J Med Sci 75: 169-79, 2013
- Ito Y, Suzuki R, Torii Y, Kawa K, Kikuta A, Kojima S, Kimura H. HLA-A*26 and HLA-B*52 are associated with a risk of developing EBV-associated T/NK lymphoproliferative disease. Blood e-Letter ID: bloodjournal_el; 8085, 2013
- Suzuki M, Torii Y, Kawada J, Kimura H, Kamei H, Onishi Y, Kaneko K, Ando H, Kiuchi T, Ito Y. Immunogenicity of inactivated seasonal influenza vaccine in adult and pediatric liver transplant recipients over two seasons. Microbiol Immunol 57:715-22.2013
- Kawano Y, Iwata S, Kawada J, Gotoh K, Suzuki M, Torii Y, Kojima S, Kimura H, Ito Y.Plasma viral MicroRNA profiles reveal potential biomarkers for chronic active Epstein-Barr virus infection.J Infect Dis 208: 771-9, 2013
- Sugimoto A, Sato Y, Kanda T, Murata T, Narita Y, Kawashima D, Kimura H, Tsurumi T.Different Distributions of Epstein-Barr virus early and late gene transcripts within viral replication compartments.J Virol 87: 6693-9, 2013
- Ito Y, Kimura H, Torii Y, Hayakawa M, Tanaka T, Tajiri H, Yoto Y, Tanaka-Taya K, Kanegane H, Nariai A, Sakata H, Tsutsumi H, Oda M, Yokota S, Morishima T, Moriuchi H. Risk factors for poor outcome in congenital cytomegalovirus infection and neonatal herpes on the basis of a nationwide survey in Japan.Pediatr Int 55, 566–571, 2013
- Trii Y, Kimura H, Ito Y, Hayakawa M, Tanaka T, Tajiri H, Yoto Y, Tanaka-Taya K, Kanegane H, Nariai A, Sakata H, Tsutsumi H, Oda M, Yokota S, Morishima T, Moriuchi H.Clinico-epidemiological states of mother-to-child infections: a nationwide survey in Japan. Pediatr Infect Dis J 32:699-701, 2013
- Murata T, Iwata S, Siddiquey NA, Kanazawa T, Goshima F, Kimura H, Tsurumi T.Heat shock protein 90 inhibitors repress latent membrane protein 1 (LMP1) expression and proliferation of Epstein-Barr virus-positive natural killer cell lymphoma.PLoS One 8:e63566, 2013
- Narita Y, Murata T, Ryo A, Kawashima D, Sugimoto A, Kanda T, Kimura H, Tsurumi T.Pin1 interacts with the Epstein-Barr virus DNA polymerase catalytic subunit and regulates viral DNA replication.J Virol 87:2120-7, 2013
- Esaki S, Goshima F, Kimura H, Murakami S, Nishiyama Y.Enhanced antitumoral activitiy of oncolytic herpes simplex virus with gemcitabine using colorectal tumor models.Int J Cancer 132:1592-601, 2013
- Isobe Y, Hamano Y, Yoshinori Ito Y, Kimura H, Tsukada N, Sugimoto K, Komatsu N. A monoclonal expansion of Epstein–Barr virus-infected natural killer cells after allogeneic peripheral blood stem cell transplantation.J Clin Virol 56:150-2, 2013
- Kawada J, Ito Y, Torii Y, Kimura H, Iwata N.Remission of Juvenile Idiopathic Arthritis with Primary Epstein-Barr Virus Infection. Rheumatol 52:956-8 2013
- Ohta R, Imai M, Kawada J, Kimura H, Ito Y.Interleukin-17A-producing T lymphocytes in chronic active Epstein-Barr virus infection.Microbiol Immunol 57:139-44, 2013
- Hiraiwa-Sofue A, Ito Y, Ohta R, Kimura H, Okumura A.Human herpesvirus 6-associated encephalopathy in a child with Dravet syndrome. Neuropediatrics 44:155-8, 2013
- Torii Y, Kimura H, Hayashi K, Suzuki M, Kawada J, Kojima S, Katano Y, Goto H, Ito Y.Causes of vertical transmission of hepatitis B virus under the at-risk prevention strategy in Japan.Microbiol Immunol 57:118-21, 2013
- Ito Y, Kawamura Y, Iwata S, Kawada J, Yoshikawa T, Kimura H. Demonstration of type II latency in T lymphocytes of Epstein-Barr Virus -associated hemophagocytic lymphohistiocytosis. Pediatr Blood Cancer 60: 326-328, 2013
2012
- Kimura H, Ito Y, Kawabe S, Gotoh K, Takahashi Y, Kojima S, Naoe T, Esaki S, Kikuta A, Sawada A, Kawa K, Ohshima K, Nakamura S. Epstein-Barr virus (EBV)-associated T/NK lymphoproliferative diseases in non-immunocompromised hosts: prospective analysis of 108 cases. Blood 119:673-86, 2012
- Ito Y, Kimura H, Maeda Y, Hashimoto C, Ishida F, Izutsu K, Sueoka E, Isobe Y, Takizawa J, Hasegawa Y, Kobayashi H, Okamura S, Kobayashi H, Yamaguchi M, Suzumiya J, Hyo R, Nakamura S, Kawa K, Oshimi K, Suzuki R. Pretreatment EBV-DNA copy number is predictive of response and toxicities to SMILE chemotherapy for extranodal NK/T-cell lymphoma, nasal type. Clin Cancer Res 18: 4183-4190, 2012
- Li Z, Yamauchi Y, Kamakura M, Murayama T, Goshima F, Kimura H, Nishiyama Y. Herpes simplex virus requires PARP activity for efficient replication and induces ERK-dependent phosphorylation and ICP0-dependent nuclear localization of tankyrase 1. J Virol 86: 492-503, 2012
- Ko Y-H, Kim H-J, Oh Y-H, Park G, Lee S-S, Huh J, Kim C-W, Kim I, Ng S-B, Tan S-Y, Chuang S-S, Nakamura N, Yoshino T, Nakamura S, Kimura H, Ohshima K. EBV-associated T and NK cell lymphoproliferative disorders: Consensus report of the 4th Asian Hematopathology Workshop. J Hematopathol 5:319–324, 2012
- Muto Y, Goshima F, Ushijima Y, Kimura H, Nishiyama Y. Generation and characterization of UL21-null herpes simplex virus type 1. Front Microbiol 3:394, 2012
- Esaki S, Yamano K, Kiguchi J, Katsumi S, Keceli S, Okamoto H, Goshima F, Kimura H, Nishiyama Y, Murakami S. Diabetic mice show an aggravated course of herpes-simplex-virus-induced facial nerve paralysis. Otol Neurotol 33:1452-7, 2012
- Sahin TT, Kasuya H, Nomura N, Shikano T, Yamamura K, Gewen T, Kanzaki A, Fujii T, Sugae T, Imai T, Nomoto S, Takeda S, Sugimoto H, Kikumori T, Kodera Y, Nishiyama Y , Nakao A. Impact of novel oncolytic virus HF10 on cellular components of the tumor microenviroment in patients with recurrent breast cancer. Cancer Gene Therapy 19:229-37,2012
- Higashimoto Y, Ohta A, Nishiyama Y , Ihira M, Sugata K, Asano Y, Peterson DL, Ablashi DV, Lusso P, Yoshikawa T. Development of a human herpesvirus 6 species-specific immunoblotting assay. Journal of Clinical Microbiology 50:1245-51,2012
- Kawabe S, Ito Y, Gotoh K, Kojima S, Matsumoto K, Kinoshita T, Iwata S, Nishiyama Y, Kimura H. Application of flow cytometric in situ hybridization assay to Epstein–Barr virus-associated T/NK lymphoproliferative diseases. Cancer Sci 103: 1481-8, 2012
- Yasuda K, Sugiura K, Ishikawa R, Kihira M, Negishi Y, Iwayama H, Ito K, Kimura H, Kosugi I, Akiyama M. Perinatal cytomegalovirus -associated bullae in an immunocompetent infant. Arch Dermatol 148: 770-2, 2012
- Luo C, Goshima F, Kamakura M, Mutoh Y, Iwata S, Kimura H, Nishiyama Y. Immunization with a highly attenuated replication -competent herpes simplex virus type 1 mutant, HF10, protects mice from genital disease caused by herpes simplex virus type 2. Front Microbiol 3: 158, 2012
- Isobe Y, Aritaka N, Setoguchi Y, Ito Y, Kimura H, Hamano Y, Sugimoto K, Komatsu N. T/NK-cell type chronic active Epstein-Barr virus (EBV) disease in adults: an underlying condition for EBV-associated T/NK-cell lymphoma. J Clin Pathol 65: 278-82, 2012
- Hirai Y, Yamamoto T, Kimura H, Ito Y, Tsuji K, Miyake T, Morizane S, Suzuki D, Fujii K, Iwatsuki K. Hydroa vacciniforme is associated with increased numbers of Epstein-Barr virus-infected T-cells. J Invest Dermatol 132:1401-8, 2012
- Iwata S, Saito T, Ito Y, Kamakura M, Gotoh K, Kawada J, Nishiyama Y, Kimura H. Antitumor activities of valproic acid on Epstein-Barr virus-associated T and natural killer lymphoma cells. Cancer Sci 103:375-8, 2012
- Kawada J, Iwata N, Kitagawa Y, Kimura H, Ito Y. Prospective monitoring of Epstein-Barr virus and other herpesviruses in patients with juvenile idiopathic arthritis treated with methotrexate and tocilizumab. Mod Rheumatol 22: 565-70, 2012

