Gastroenterological Surgery
KEYWORDS
- GI Cancer: Pathophysiology
- Clinical Res
HEAD

LAB MEMBER
| Faculty | Position | Researchers |
|---|---|---|
| TANAKA Chie | Clinical Associate Professor | Researchers |
| HAYASHI Masamichi | Lecturer | Researchers |
| HATTORI Norifumi | Clinical Lecturer | Researchers |
| TAKAMI Hideki | Clinical Lecturer | Researchers |
| SHIMIZU Dai | Clinical Lecturer | Researchers |
| SATO Yusuke | Clinical Lecturer | Researchers |
| TANAKA Haruyoshi | Clinical Lecturer | Researchers |
| NAKANISHI Koki | Clinical Lecturer | Researchers |
| UMEDA Shinichi | Assistant Professor | Researchers |
| KURIMOTO Keisuke | Clinical Assistant Professor | Researchers |
| NAKAGAWA Nobuhiko | Clinical Assistant Professor | Researchers |
| IIZUKA Akimitsu | Clinical Assistant Professor | Researchers |
CONTACT
| iga-ryu◎t.mail.nagoya-u.ac.jp (Please send a message after replacing "◎" mark with "@" mark. ) | |
| HP | Private Page |
OUTLINE
Training Outstanding Surgeons and Sharing Excellence with the World Through Both Clinical Practice and Research
Our department is committed to training surgeons in gastrointestinal surgery who possess both solid clinical competence and advanced research capability. In clinical practice, we faithfully inherit our long-standing tradition of fundamental principles in cancer surgery while implementing state-of-the-art, evidence-based surgical care across the full spectrum—from function-preserving and minimally invasive procedures to extended radical resections.
In research, we actively promote both basic and clinical studies aimed at developing new therapeutic strategies, with a primary focus on gastrointestinal cancers and inflammatory bowel disease. Our efforts include biomarker discovery and drug development research. By integrating extensive clinical experience with the latest scientific knowledge, we generate research outcomes that can be disseminated globally and place strong emphasis on nurturing the next generation of surgeons.
In recent years, the nationwide decline in the number of surgeons has become a serious social issue. To address this proactively, we are developing a structured training environment centered on laparoscopic and robot-assisted surgery. In addition, we are strengthening educational systems that support the continuous growth of early-career surgeons and building a workplace in which diverse talent—including female physicians—can thrive over the long term.
Surgeons are expected not only to master exceptional operative technique, but also to possess broad medical knowledge, comprehensive clinical judgment, and the ethics and humanity required to engage sincerely with critically ill patients and their families and to confront disease together as a team. We aim to pass on to young physicians who will lead the future of healthcare the sense of accomplishment and pride that comes with the responsibilities of surgery, along with the necessary knowledge and technical skills. Working closely with our affiliated hospitals, we prioritize the acquisition of balanced, comprehensive clinical ability, with certification as a board-certified surgeon as the initial milestone. Thereafter, we provide an environment in which trainees can gain extensive case experience to achieve board certification in gastrointestinal surgery at the earliest possible stage. To foster surgeons with a broad perspective not confined to a single subspecialty, we also maintain a training system that enables learning from as many supervising surgeons as possible.
Building on these educational and clinical foundations, we further encourage the pursuit of advanced expertise through graduate-level training in the Graduate School of Medicine and aim to expand both basic and clinical research that can be communicated nationally and internationally. By integrating the surgical traditions inherited from our predecessors with cutting-edge medicine, our shared departmental goal is to “train outstanding surgeons and share excellence with the world through both clinical practice and research.
RESEARCH PROJECTS
1. Esophageal Group
- Comprehensive screening of cancer-associated genes in esophageal cancer and functional analyses of their expression
- Study evaluating the efficacy and safety of robot-assisted esophagectomy using the da Vinci Surgical System for esophageal cancer
- Phase II clinical trial to evaluate the efficacy and safety of preoperative docetaxel + oxaliplatin + S-1 therapy for advanced esophageal cancer (CCOG2102)
- Study on the efficacy of immediate preoperative nutritional intervention following neoadjuvant chemotherapy in patients with esophageal cancer (CCOG2103)
- Study of oscillatory positive expiratory pressure (OPEP) therapy during the perioperative period for esophageal cancer (CCOG2302)
- Randomized controlled trial investigating omission of prophylactic supraclavicular lymph node dissection for thoracic upper/middle esophageal squamous cell carcinoma, clinical stage I–IVA (excluding T4) (JCOG2013)
- Randomized phase II/III trial of neoadjuvant chemotherapy with DOS or FLOT for adenocarcinoma of the esophagogastric junction (JCOG2203)
- Randomized phase III trial comparing postoperative observation (no adjuvant therapy) vs nivolumab vs S-1 in patients with esophageal squamous cell carcinoma who underwent curative surgery after neoadjuvant chemotherapy without achieving a pathological complete response (JCOG2206)
2. Gastric Group
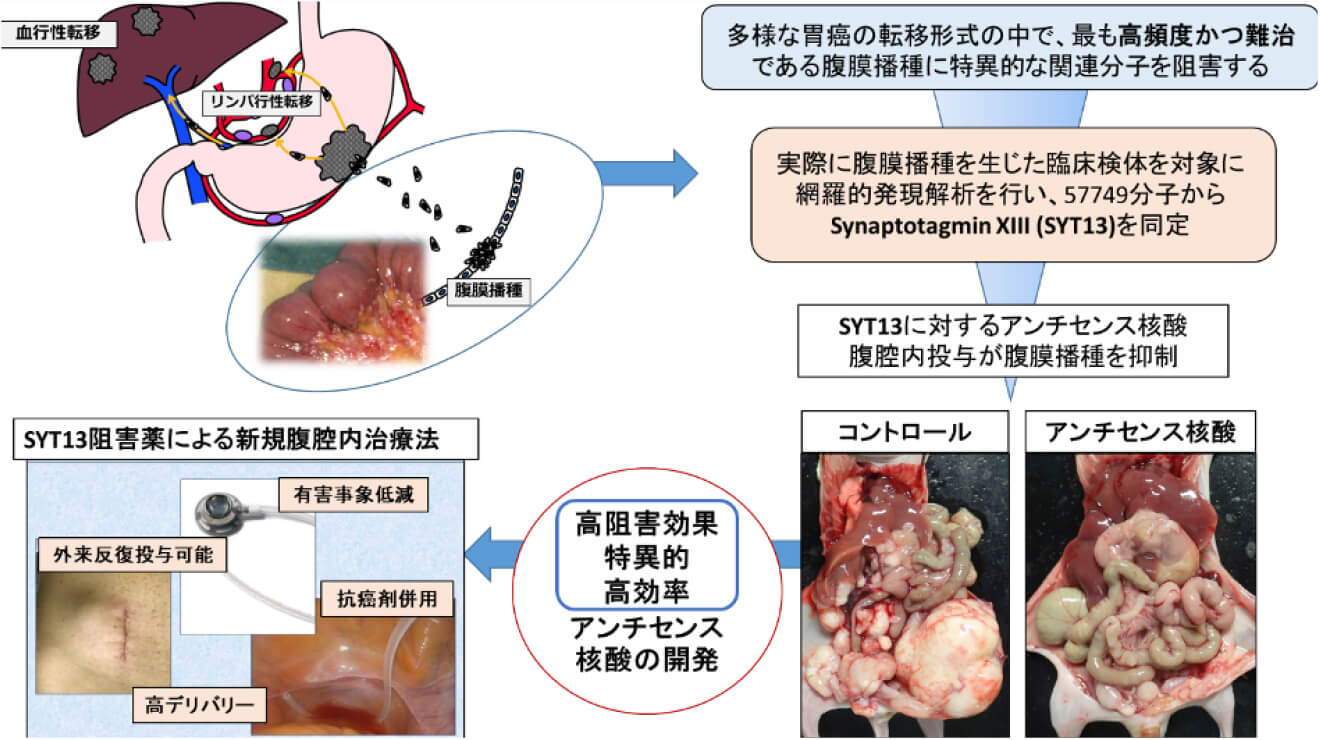
1) Development of a novel molecularly targeted therapy specific to peritoneal metastasis of gastric cancer
Peritoneal metastasis is a common mode of recurrence and dissemination in gastric cancer and remains highly treatment-resistant, representing a major determinant of prognosis. Our department has conducted consistent translational research—from basic biology to drug development—focusing on SYT13, a molecule that is highly expressed specifically in gastric cancer peritoneal metastases and plays a pivotal role in their formation and progression. We have developed ASO-4733, an antisense oligonucleotide therapeutic incorporating amido-bridged nucleic acids targeting SYT13, and have accumulated non-clinical data demonstrating both efficacy and safety in peritoneal metastasis models. Building on these achievements, we are currently conducting an investigator-initiated Phase I clinical trial in patients with unresectable advanced or recurrent gastric cancer with peritoneal metastasis, to validate the clinical feasibility of intraperitoneal administration as a new therapeutic strategy.
2) Transcriptome-based discovery of liver metastasis–specific biomarkers in gastric cancer according to metastatic patterns
Hematogenous metastasis of gastric cancer, exemplified by liver metastasis, is known to develop through a multistep process involving numerous molecules. We hypothesize that certain molecules are specific to distinct metastatic routes. We therefore aim to identify molecules that show aberrant expression specifically in cases with liver metastasis or recurrence through comprehensive gene expression analyses, and to evaluate their potential as diagnostic and predictive biomarkers for gastric cancer liver metastasis.
3) Development of a novel marking method for early gastric cancer using indocyanine green (ICG)
4) Functional assessment of older adults undergoing gastric cancer surgery
5) Development of an integrated basic task program for endoscopic surgical simulation training
6) Randomized controlled trial evaluating the impact of anamorelin hydrochloride on the relative dose intensity of adjuvant chemotherapy in patients after gastrectomy for gastric cancer
7) International prospective validation of a serum tumor marker assay kit for screening, diagnosis, staging, response assessment, and postoperative recurrence monitoring
There is a strong demand for serum tumor markers that can be obtained non-invasively and easily, across all clinical settings including population-based screening, cancer diagnosis, staging for treatment decision-making, assessment of treatment response, and monitoring for recurrence after curative resection. Our department has established an assay kit for a serum marker discovered in-house and is conducting an international prospective collaborative study with partners in Korea and the United States to validate its clinical utility.
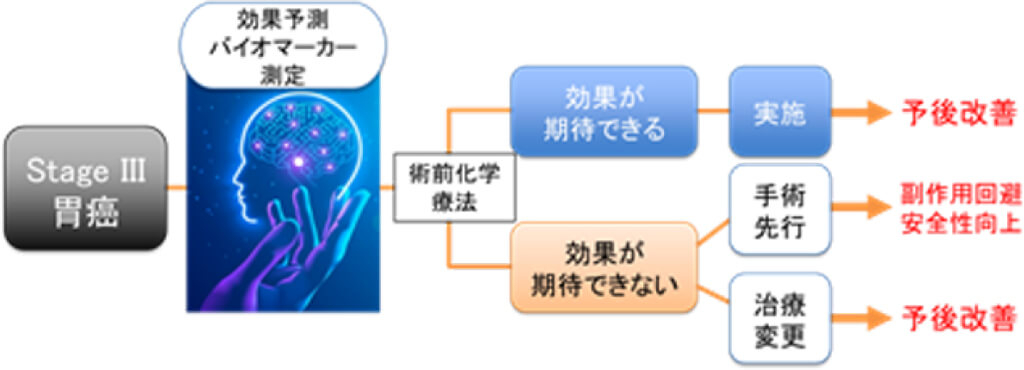
8) Development of predictive biomarkers for response to neoadjuvant chemotherapy in advanced gastric cancer
Using blood and tissue specimens, we aim to identify high-performance and highly novel predictive biomarkers for response to neoadjuvant chemotherapy in advanced gastric cancer by applying AI-driven integrative analyses to multi-omics data.
3. Hepatic Group
1) Identification of methylated genes presumed to contribute to the progression of hepatocellular carcinoma
When sequencing bisulfite-treated samples of hepatocellular carcinoma cell lines, the CpG sites in Hep3B (moderately differentiated carcinoma) were TG (unmethylated), whereas in HLE (undifferentiated carcinoma), the CpG sites had changed to CG (methylated). In resected hepatocellular carcinoma specimens, cases with methylation-positive status (red line) had significantly poorer overall postoperative survival compared to methylation-negative cases (blue line) (BMC Cancer, 2014).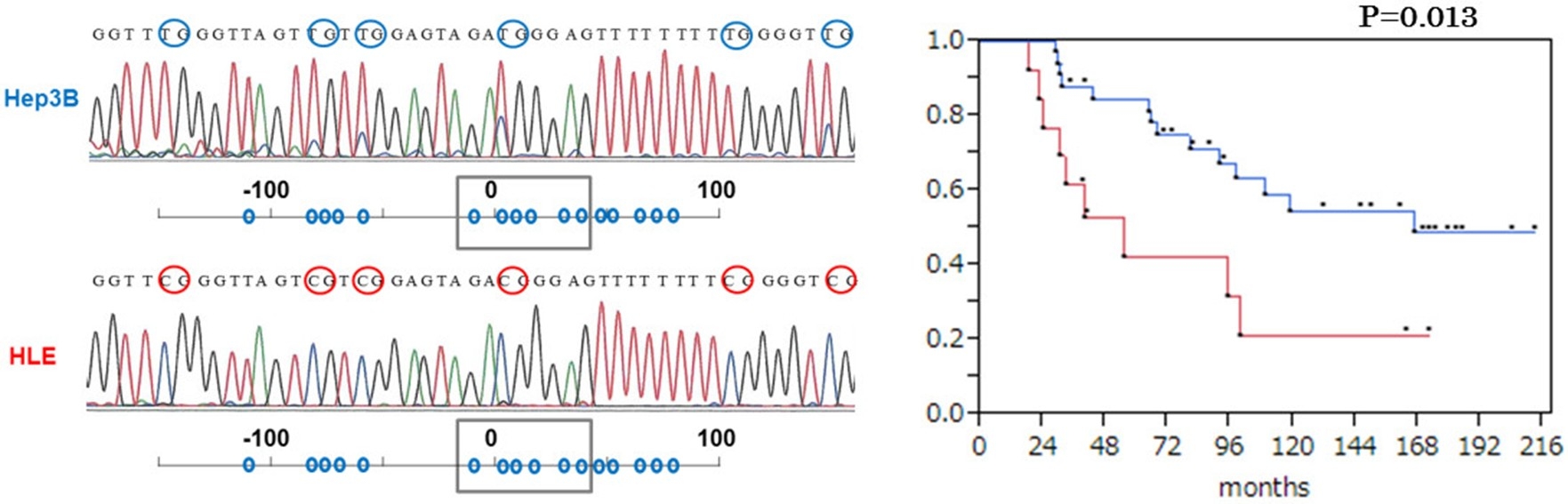
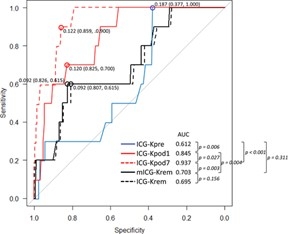
2) Perioperative measurement of ICG-K values and prediction of postoperative liver failure in hepatectomy cases without biliary reconstruction
In addition to preoperative ICG-K value (ICG-Kpre) and postoperative remnant liver ICG-K value (mICG-Krem), ICG-K values on postoperative day 1 (ICG-Kpod1) and day 7 (ICG-Kpod7) were measured, and their correlation with postoperative liver failure (Grade B/C) was evaluated. mICG-Krem correlated well with ICG-Kpod1 and ICG-Kpod7. On multivariate analysis, ICG-Kpod7 and postoperative bile leakage were significant predictors of liver failure (J Hepatobiliary Pancreatic Sci, 2021).
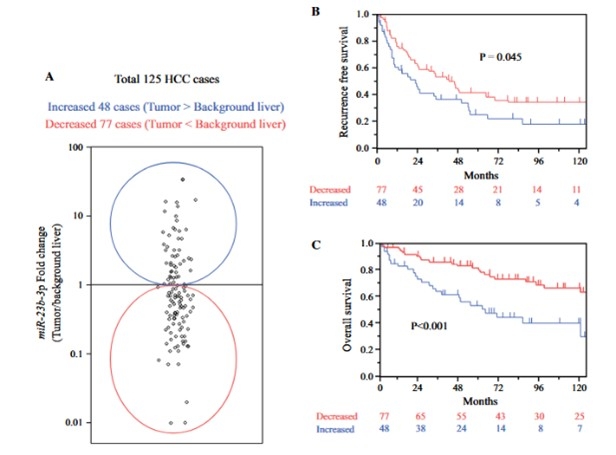
3) Increased expression of miR-23b in hepatocellular carcinoma tissue is a poor prognostic factor after hepatectomy
In hepatocellular carcinoma, cases with increased expression of miR-23b in tumor tissue compared to the background liver showed significantly poorer postoperative prognosis after hepatectomy. Upregulation of miR-23b has been shown to reduce the expression of genes such as MICU3 and AUH, which are involved in maintaining mitochondrial function, and is thought to contribute to cancer cell malignancy (Ann Surg Oncol, 2021).
4. Pancreatic Group
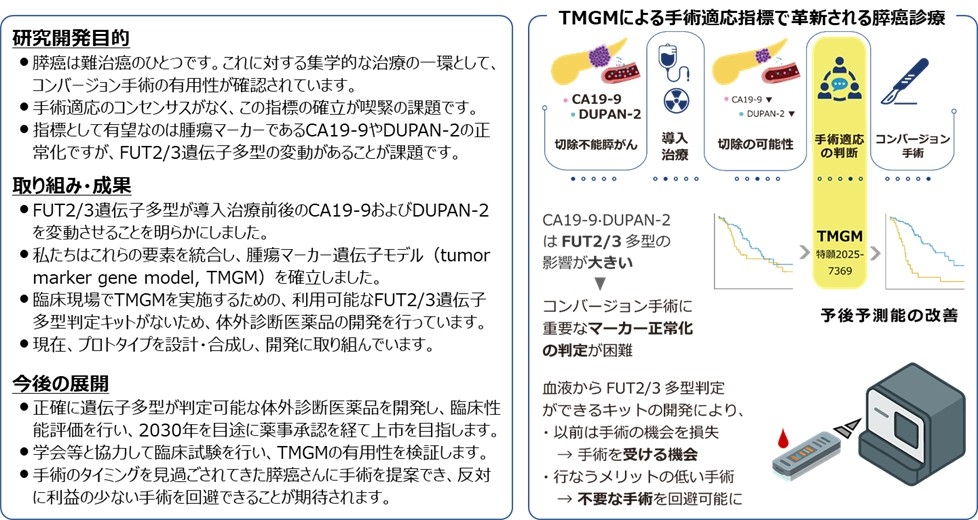
1) Development of a Tumor Marker Gene Model for Advanced Pancreatic Cancer
For advanced pancreatic cancer, timely surgery as part of multidisciplinary treatment is crucial. Tumor markers, particularly CA19-9 and DUPAN-2, are useful references for determining surgical feasibility, but their individual variability has been a challenge. We developed a Tumor Marker Gene Model (TMGM) combining CA19-9, DUPAN-2, and FUT2/FUT3 gene polymorphisms, which demonstrated high prognostic predictive ability. We are currently developing a FUT2/3 gene polymorphism detection kit to clinically apply TMGM as a reference model for determining surgical eligibility.
Tanaka H, Sakai A, Suenaga M, et al. FUT2 and FUT3-specific normalization of DUPAN-2 and carbohydrate antigen 19-9 in preoperative therapy for pancreatic cancer: multicentre retrospective study (GEMINI-PC-01). Br J Surg. 2025;112:znaf049.
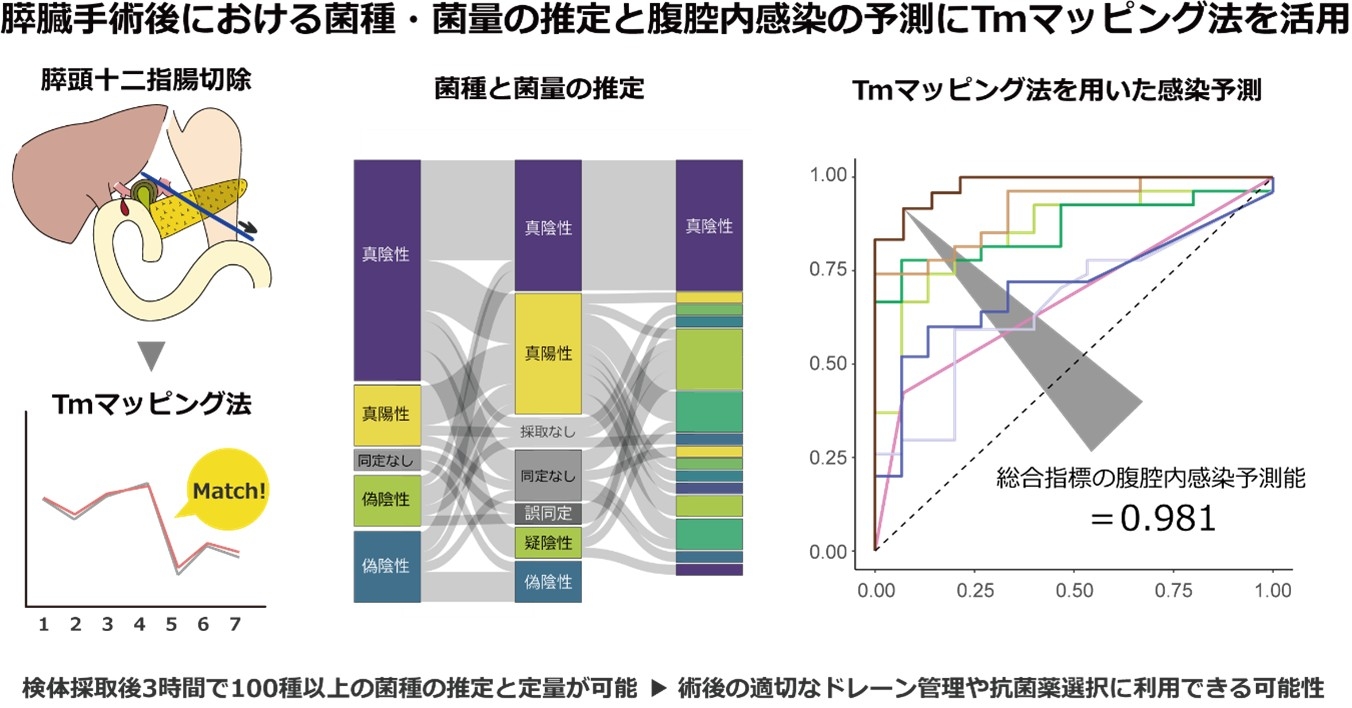
2)Research on Safe Perioperative Infection Control for Pancreatoduodenectomy
Our department is conducting joint research with the University of Toyama to develop and apply a novel rapid bacterial testing technology for predicting postoperative infections. Pancreatoduodenectomy (PD) is one of the most demanding gastrointestinal surgeries, and postoperative intra-abdominal infection remains a significant challenge. Controlling postoperative intra-abdominal infections after pancreatectomy remains a critical challenge. Conventional bacterial culture and Gram staining tests have limitations in speed and detail. The new “Tm mapping method” estimates the “type and quantity” of causative bacteria in as little as approximately 3 hours. We validated its usefulness in patients after pancreaticoduodenectomy. The AUC of the predictive model including Tm mapping was 0.981, suggesting potential for improving the quality of postoperative management decisions.
Tanaka H, Fukasawa M, Kimura N, et al. A novel melting temperature mapping method may improve the prediction of postoperative intra-abdominal infection after pancreatoduodenectomy. Br J Surg. 2025 Oct 3;112:znaf222. 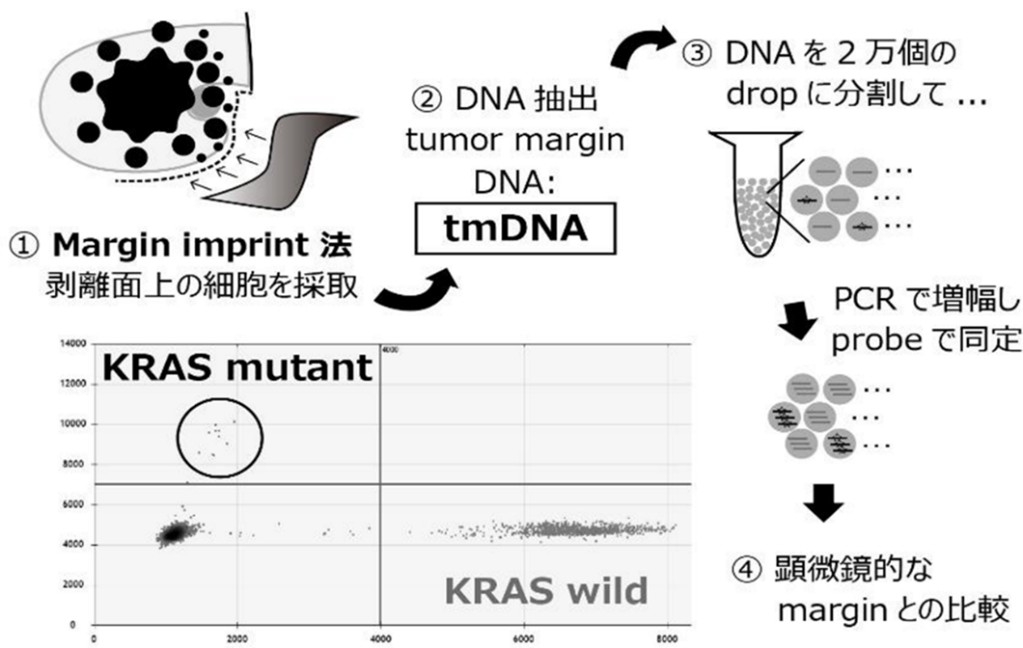
3)Development of diagnostics for pancreatic cancer's molecular resection margin status at dissected peripancreatic tissue margin by margin imprint droplet digital PCR (PIRAMID study)
Pancreatic cancer is one of the most challenging cancers to control. Even when macroscopically complete resection is achieved, microscopic residual disease may sometimes be present. Furthermore, even when pathologically diagnosed as completely resected, local recurrence may occur after radical resection. To overcome this challenge, we are attempting a molecular biological approach to detect residual cancer during pancreatic cancer resection. We have established a margin imprint digital PCR method to molecularly diagnose the presence of cancer on the surgical resection margin of pancreatic cancer. By applying an imprint membrane to the surgical dissection margin of freshly resected pancreatic cancer specimens, we efficiently collect cells from the irregular, extensive surgical margin. We then use nucleic acids extracted from these cells to attempt molecular biological identification of ultra-low-frequency cancer cells. In the future, we aim to develop this as an in vitro diagnostic medical device for predicting postoperative intraperitoneal recurrence in clinical practice.
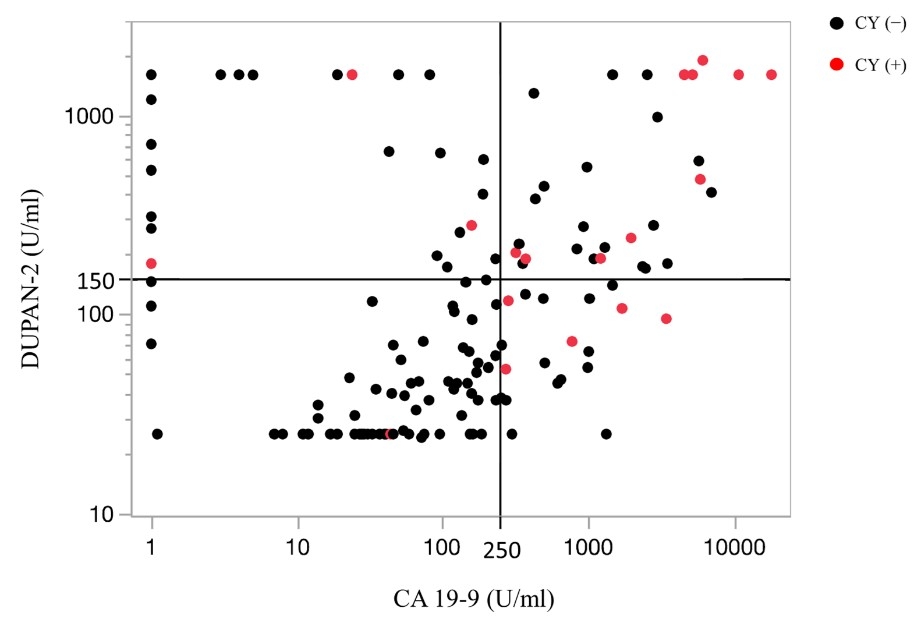
4) One factor influencing pancreatic cancer prognosis is positive peritoneal cytology—a condition where pancreatic cancer cells are present in the abdominal cavity—which is considered a poor prognostic factor. We demonstrated that pancreatic cancer patients with positive peritoneal cytology frequently exhibit CA19-9 ≥250 U/mL, DUPAN-2 ≥150 U/mL, and are more likely to have resectable margins.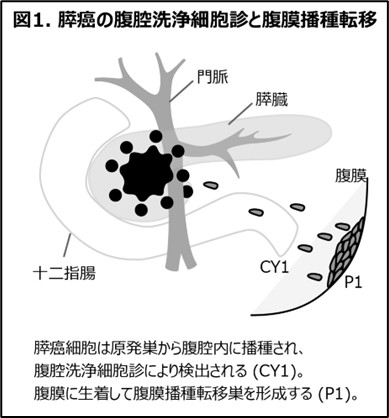
5) Accompanying study of a multicenter phase II study to investigate the efficacy of systemic chemotherapy in patients with peritoneal washing cytology-positive potentially curable pancreatic cancer
Validation of quantifying peritoneal tumor DNA during systemic chemotherapy to monitor negative conversion of cancerous cytology and exploration for novel biomarkers of pancreatic cancer (WALCURE DRIVE): A factor influencing pancreatic cancer prognosis is positive peritoneal cytology, meaning pancreatic cancer cells are circulating in the peritoneal cavity. We are validating the efficacy of a strategy involving preoperative treatment until this becomes negative, with Nagoya University actively participating (WALCURE trial). This is an ongoing clinical trial and is led by the University of Toyama. We are responsible for a accompanying study of the WALCURE trial, which verifies this treatment efficacy from a molecular biological perspective, such as using digital PCR.
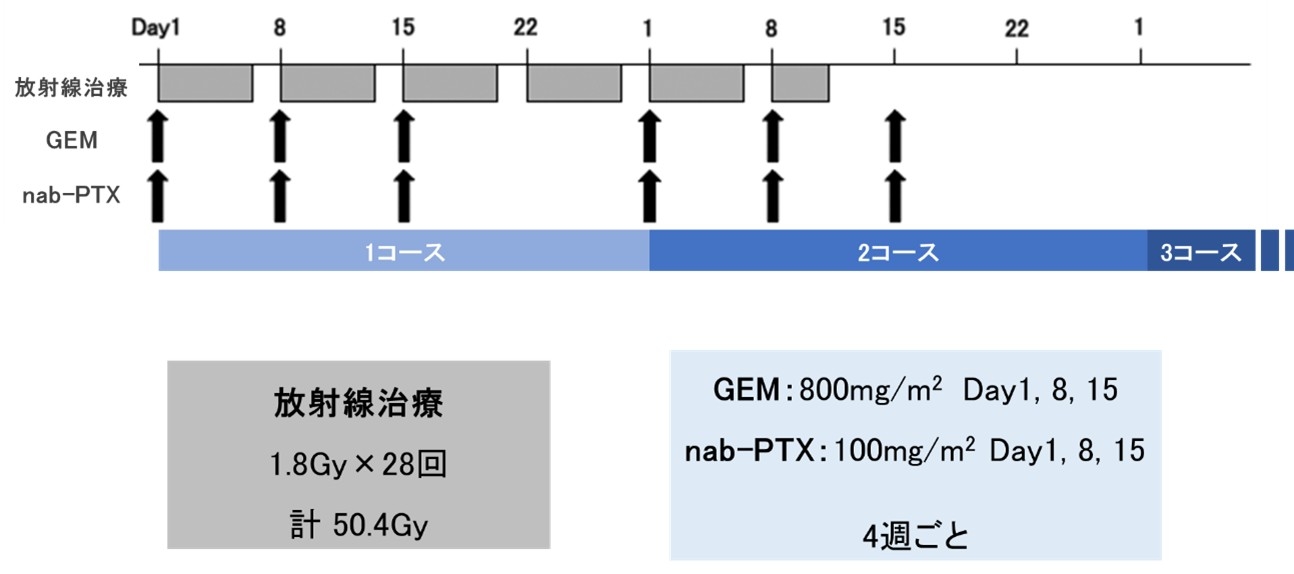
6) Phase I study of chemoradiotherapy using gemcitabine plus nab-paclitaxel for unresectable locally advanced pancreatic cancer
A physician-initiated Phase I trial was conducted to determine the recommended therapeutic dose for chemoradiotherapy (GnP-CRT), combining gemcitabine and nab-paclitaxel—a regimen gaining attention in recent years—with radiotherapy for locally advanced pancreatic cancer without distant metastasis. Yamada S, Fujii T, Yokoyama Y, Kawashima H, Maeda O, Suzuki K, Okada T, Ono E, Yamaguchi J, Takano N, Takami H, Hayashi M, Niwa Y, Hirooka Y, Ito Y, Naganawa S, Ando Y, Nagino M, Goto H, Kodera Y. Cancer Chemother Pharmacol. 2018;81:815-821.
7) Phase II study of chemoradiotherapy combined with gemcitabine and nab-paclitaxel for unresectable locally advanced pancreatic cancer (NUPAT-05)
This clinical trial aimed to improve treatment outcomes for locally advanced pancreatic cancer without distant metastasis. It combined the recently noted gemcitabine and nab-paclitaxel therapy with radiotherapy to enhance therapeutic efficacy and further explored conversion surgery to improve treatment results. We are currently compiling these treatment outcomes and preparing for publication in an academic journal.
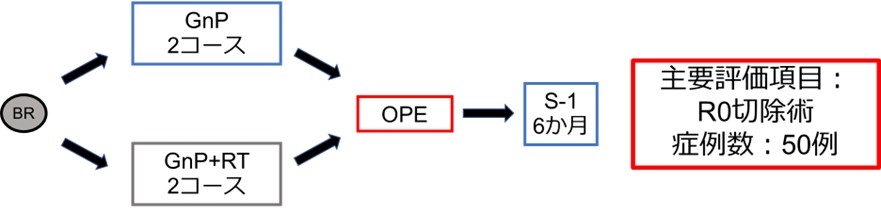
8) Randomized Phase II Clinical Trial of Preoperative nab-PTX+GEM Therapy ± Radiotherapy for Borderline Resectable Pancreatic Cancer; Nagoya University Pancreatic Tumor Board trial 07 (NUPAT-07)
Gemcitabine and nab-paclitaxel therapy is widely used at institutions treating pancreatic cancer where surgical resection is challenging (borderline resectable pancreatic cancer). A clinical trial is underway to investigate the additive effect of radiation therapy by randomly assigning patients to receive either nab-PTX+GEM therapy alone or nab-PTX+GEM therapy plus radiation therapy, followed by surgery. [Figure: Pancreas ⑦]
9) Randomized Phase II Clinical Trial on the Timing of Radiotherapy Introduction for Locally Advanced Pancreatic Cancer
Nagoya University Pancreatic Tumor Board trial 08 (NUPAT-08): As described above, the NUPAT-05 trial was conducted as a clinical trial combining gemcitabine and nab-paclitaxel therapy with radiotherapy for pancreatic cancer with a locally advanced unresectable pancreatic cancer, yielding promising treatment outcomes. However, concerns have been raised that the interval between radiotherapy and surgery may increase the difficulty of the surgery itself. Therefore, a clinical trial is underway to compare the treatment regimen used in NUPAT-05 with a regimen that delays the timing of radiotherapy, aiming to determine the optimal multidisciplinary treatment strategy. 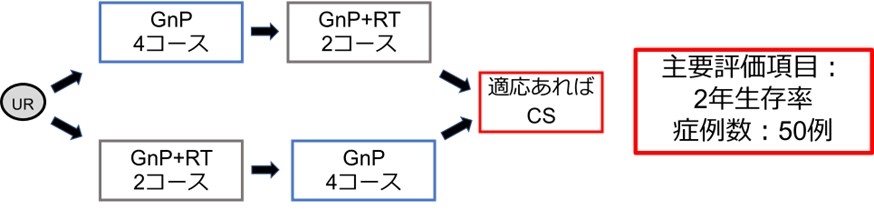
5. Colorectal Group
Colorectal Cancer
1) Optimization of Chemotherapy for Advanced/Recurrent Colorectal Cancer
Phase II study of FOLFIRI with low-dose irinotecan plus ramucirumab as second-line treatment in Japanese patients with metastatic colorectal cancer (RINDO study)
We investigated the efficacy and safety of FOLFIRI plus ramucirumab using the Japanese standard dose of irinotecan (150 mg/m²) as second-line chemotherapy for metastatic colorectal cancer, and published the findings.
- Jpn J Clin Oncol. 2025 Dec 8; hyaf198. PMID: 41359440
2) Optimization of Postoperative Adjuvant Therapy in High-Risk Stage II/Stage III Colorectal Cancer (CCOG1302)
A randomized phase II trial comparing intermittent versus continuous oxaliplatin administration in CAPOX therapy for high-risk stage II/III colorectal cancer was conducted, and a subgroup analysis in elderly patients was performed.
In elderly patients, persistent peripheral neuropathy was significantly more frequent in the continuous administration group.
- Int J Clin Oncol. 2025; 30(6):1174–1182.
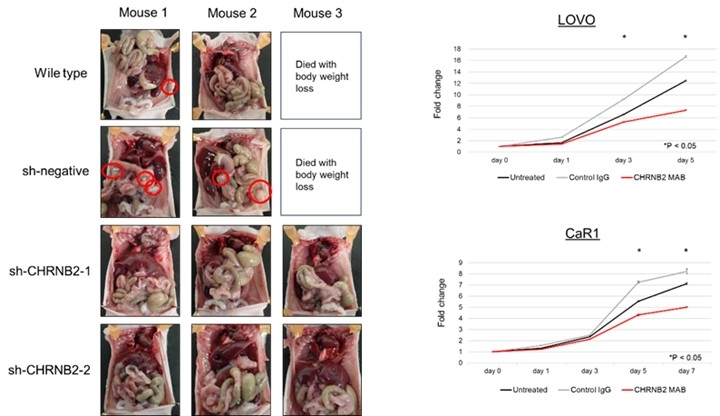
3) Identification and Functional Analysis of a Peritoneal Metastasis–Related Molecule: CHRNB2
Using resected colorectal cancer specimens, we demonstrated that patients with high CHRNB2 expression exhibited a specifically higher rate of peritoneal metastasis. Further analyses using cell lines and a mouse peritoneal metastasis model revealed that CHRNB2 inhibition or antibody treatment suppressed peritoneal dissemination and tumor growth, indicating its potential as a therapeutic target.
- Cancers (Basel). 2025; 17(15):2485.
4) Exploration of Novel Prognostic Biomarkers in Colorectal Cancer (Ongoing)
Analyses using colorectal cancer resection specimens and cell lines have suggested that GABRD, SLC7A9, FTCD, and TTYH1 may correlate with prognosis. Functional analyses and clinical validation studies are currently underway.
Inflammatory Bowel Disease (IBD)
1) Incidence and Risk Factors of Portal Vein Thrombosis After IBD Surgery (Ongoing)
A prospective observational study is being conducted to evaluate the incidence and risk factors of venous thromboembolism, including portal vein thrombosis, by performing contrast-enhanced CT one week after surgery for ulcerative colitis and Crohn’s disease.
2) Phase III Trial of Guselkumab for Perianal Fistulizing Crohn’s Disease
We are participating in a multicenter phase III clinical trial evaluating the efficacy and safety of guselkumab in patients with Crohn’s disease complicated by perianal fistula.
3) Evaluation of Triangular Anastomosis for Crohn’s Disease Bowel Resection (CCOG2503)
A prospective study is underway to test the hypothesis that triangular end-to-end anastomosis using a linear stapler decreases anastomotic leakage and improves postoperative endoscopic passage in Crohn’s disease bowel resection.
4) Evaluation of the Preventive Effect of Saireito on High-Output Stoma in Temporary Ileostomy Patients with Ulcerative Colitis
Based on previous findings in covering ileostomy for colorectal cancer, a clinical study is currently examining whether the Japanese herbal medicine Saireito can prevent high-output stoma (HOS) in ulcerative colitis patients with temporary ileostomy after surgery.
5) Comprehensive Analysis of Drug Resistance and Carcinogenesis-Related Genes in Ulcerative Colitis (Ongoing)
Using resected ulcerative colitis specimens, comprehensive mRNA expression analyses are being performed on:
- infliximab-resistant mucosa
- colitic cancer (cancerous and non-cancerous areas)
- sporadic cancer (cancerous and non-cancerous areas)
(10 cases each)
This project aims to identify genes involved in drug resistance and carcinogenesis in ulcerative colitis.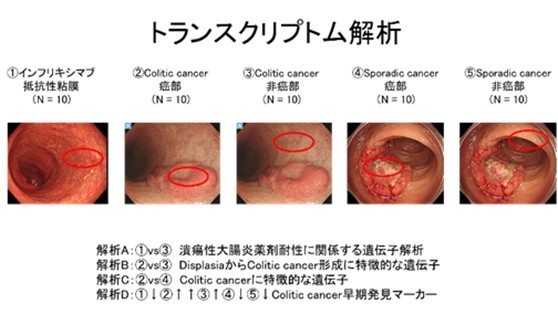
BIBLIOGRAPHY
2024 (Clinical)
- Nakanishi, K. Kanda, M. Ito, S. Mochizuki, Y. Teramoto, H. Ishigure, K. Murai, T. Asada, T. Ishiyama, A. Matsushita, H. Shimizu, D. Tanaka, C. Fujiwara, M. Murotani, K. Kodera, Y. Oncological similarities between large type 3 and type 4 tumors in. patients with resectable gastric cancer: a propensity score-matched. analysis of a multi-institutional dataset. GASTRIC CANCER, 2024; 27: 1331-1341
- Kawakami, H. Nakanishi, K. Makiyama, A. Konishi, H. Morita, S. Narita, Y. Sugimoto, N. Minashi, K. Imano, M. Inamoto, R. Kodera, Y. Kume, H. Yamaguchi, K. Hashimoto, W. Muro, K. Real-world effectiveness and safety of trastuzumab-deruxtecan in. Japanese patients with HER2-positive advanced gastric cancer (EN-DEAVOR. study). GASTRIC CANCER, 2025; 28: 51-62
- Kodera, Y. The Magic Flute is a Singspiel, not an opera? My thoughts about the. German, Austrian and Swiss Guidelines for Systemic Treatment of Gastric. Cancer. GASTRIC CANCER, 2024; 27: 1-2
- Kobayashi, D. Kodera, Y. Fukushima, R. Morita, M. Fushida, S. Yamashita, N. Yoshikawa, K. Ueda, S. Yabusaki, H. Kusumoto, T. Arigami, T. Hidemura, A. Omori, T. Yamaguchi, H. Hirono, Y. Tsuji, Y. Moon, JH. Tomita, T. Imamura, H. Nakanishi, K. Shimizu, D. Hirakawa, A. Ishigami, H. Kitayama, J. Phase II Study of Intraperitoneal Administration of Paclitaxel Combined. with S-1 and Cisplatin for Gastric Cancer with Peritoneal Metastasis. ANNALS OF SURGICAL ONCOLOGY, 2024; 31: 1319-1327
- Sugiyama, F. Kanda, M. Shimizu, D. Umeda, S. Inokawa, Y. Hattori, N. Hayashi, M. Tanaka, C. Nakayama, G. Kodera, Y. Absence of Hypercoagulation Status after Neoadjuvant Treatment is. Associated with Favorable Prognosis in Patients Undergoing Subtotal. Esophagectomy for Esophageal Squamous Cell Carcinoma. ANNALS OF SURGICAL ONCOLOGY, 2024; 31: 3417-3425
2024 (Experimental)
- Kanda, M. Kasahara, Y. Shimizu, D. Shinozuka, T. Sasahara, M. Nakamura, S. Iguchi, Y. Katsuno, M. Kodera, Y. Obika, S. Dual-modified antisense oligonucleotides targeting oncogenic. protocadherin to treat gastric cancer. BRITISH JOURNAL OF CANCER, 2024; 131: 1555-1566
- Ito, Y. Kanda, M. Sasahara, M. Tanaka, C. Shimizu, D. Umeda, S. Inokawa, Y. Hattori, N. Hayashi, M. Nakayama, G. Kodera, Y. Killer cell lectin-like receptor G2 facilitates aggressive phenotypes of. gastric cancer cells via dual activation of the ERK1/2 and JAK/STAT. pathways. GASTRIC CANCER, 2024; 27: 506-518
- Kanda, M. Takano, N. Miyauchi, H. Ueda, K. Mizuno, M. Kasahara, Y. Kodera, Y. Obika, S. Preclinical toxicological assessment of amido-bridged nucleic. acid-modified antisense oligonucleotides targeting synaptotagmin XIII. for intra-abdominal treatment of peritoneal metastasis of gastric cancer. GASTRIC CANCER, 2024; 27: 1229-1241
- Sasahara, M. Kanda, M. Tanaka, C. Shimizu, D. Umeda, S. Takami, H. Inokawa, Y. Hattori, N. Hayashi, M. Nakayama, G. Kodera, Y. Therapeutic antibody targeting natriuretic peptide receptor 1 inhibits. gastric cancer growth via BCL-2-mediated intrinsic apoptosis. INTERNATIONAL JOURNAL OF CANCER, 2024; 154: 1272-1284
- Shinozuka, T. Kanda, M. Sato, Y. Shimizu, D. Umeda, S. Takami, H. Hattori, N. Hayashi, M. Tanaka, C. Kodera, Y. Therapeutic Potential of Antibody Targeting Neuronal Pentraxin Receptor. in Esophageal Squamous Cell Carcinoma. ANNALS OF SURGICAL ONCOLOGY, 2024; 31: 5674-5682

