High-Technology Application of Medicine Clinical Oncology and Chemotherapy
KEYWORDS
- Clinical Oncology
- Clinical Pharmacology
- Geriatric Oncology
- Palliative Care
HEAD

LAB MEMBER
| Faculty | Position | Researchers |
|---|---|---|
| MAEDA Osamu | Associate Professor | Researchers |
| MITSUMA Ayako | Lecturer | Researchers |
| YANAGAWA Madoka | Lecturer | Researchers |
| SHIMOKATA Tomoya | Lecturer | Researchers |
| TAKANO Yuko | Asistant Professor | Researchers |
| KONDO Chiaki | Asistant Professor | Researchers |
| MIZUNO Kazuyuki | Asistant Professor | Researchers |
| MOGI Kazumasa | Asistant Professor | Researchers |
| TSUBOI Rie | Asistant Professor | Researchers |
| MIYAI Yuki | Asistant Professor | Researchers |
CONTACT
| iga-ryu◎t.mail.nagoya-u.ac.jp (Please send a message after replacing "◎" mark with "@" mark. ) | |
| HP | Private Page |
OUTLINE
The Department of Clinical Oncology and Chemotherapy, established in 2005, is a clinical department that performs high-level outpatient pharmacotherapy for malignant neoplasms in all organs, based on close collaboration with each of the other clinical departments. The department is regularly consulted by other clinical departments on matters related to chemotherapy and palliative care.
Treatment with antineoplastic agents requires more than just familiarity with anticancer drugs and knowledge of protocols. In addition to an understanding of the clinical pharmacology of each drug and its series of adverse effects, high-level cancer pharmacology demands intimate knowledge of the pathophysiology of malignant neoplasms that occur specifically in each organ. It also demands advanced clinical skill in judging the advantages and disadvantages of various regimens depending on the response to treatment and changes in condition of individual patients.
As a result of the enthusiasm for early-phase clinical trials of anticancer agents under development, the department has been entrusted with industry-sponsored clinical trials for registration, including investigating agents that had never been administered in Japanese subjects.
RESEARCH PROJECTS
・A specified clinical research study of administering long-acting G-CSF on day 3 during neoadjuvant chemotherapy for esophageal cancer
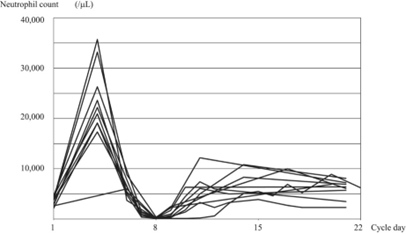
This is a clinical trial that investigated whether the prophylactic use of G-CSF, which increases neutrophil counts, reduces the risk of febrile neutropenia during preoperative chemotherapy for esophageal cancer. The figure shows the changes in neutrophil counts.
Maeda O et al. Cancer Medicine 2024
・Genomic profiles of patients with skin melanoma
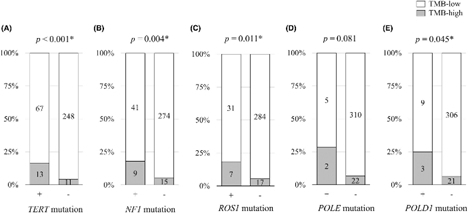
This study investigated the associations between genomic alterations in cancer cells and metastatic sites, as well as the efficacy of immune checkpoint inhibitors, using a large-scale database.
Liang Y et al. Cancer Science 2025
1. Appropriate anticancer agent therapy based on clinical pharmacology
We study the appropriate cancer pharmacotherapy for the patients requiring special consideration, including patients with organ dysfunction, elderly patients and patients with sarcopenia.
2. Individualized anticancer agent therapy based on pharmacogenetics
We study the association between various genetic polymorphism and effect or side effect of the cancer pharmacotherapy.
3. Early detection of anticancer drug’s side effect
We conduct exploratory clinical studies to discover side effects of anticancer drugs at an early stage.
4. Clinical trials in palliative medicine
We work on creation of the evidence in palliative medicine including a placebo-controlled randomized trial.
5. Early development trial of new anticancer drug
We perform the early development trial of new anticancer drugs such as molecular target agents, immune checkpoint inhibitors and so on.
BIBLIOGRAPHY
2025
- Kazuki Nishida, Yao Liang, Osamu Maeda, Angélique Da Silva, Yuichi Ando, Basile Chretien. Pazopanib and antacids: insights from the WHO pharmacovigilance database. Cancer Chemotherapy and Pharmacology. 2025 Jul 2;95(1):66. doi:10.1007/s00280-025-04792-7. PMID: 40601049.
- Kazuyuki Mizuno , Takanori Ito , Tsunaki Sawada , Tomoko Kobayashi , Shintaro Iwama , Shoichiro Mori , Tetsunari Hase , Yuki Fukami , Kenji Furusawa , Yoshimitsu Yura , Ryota Morimoto , Ai Fujita Sajiki , Hiroaki Ushida , Noritoshi Kato , Shoichi Maruyama , Toyoaki Murohara , Masahisa Katsuno , Makoto Ishii , Masashi Akiyama , Hiroshi Arima , Hiroki Kawashima , Yuichi Ando. Safety and efficacy of retreatment with immune checkpoint inhibitors after severe immune-related adverse events. Oncologist. 2025 Jun 4;30(6):oyaf120. doi: 10.1093/oncolo/oyaf120. PMID: 40515478; PMCID: PMC12166119.
- Yuko Takano, Kazuyuki Mizuno, Madoka Iwase, Sachi Morita, Nao Torii, Toyone kikumori, Yuichi Ando. \"Tumor mutational burden status and clinical characteristics of invasive lobular carcinoma of the breast. Breast Cancer. 2025 Jul;32(4):816-825.doi: 10.1007/s12282-025-01706-6. Epub 2025 May 2. PMID: 40314919
- Yao Liang, Osamu Maeda, Kazuki Nishida, Basile Chretien, Yuichi Ando. Genomic profiles of patients with skin melanoma in the era of immune checkpoint inhibitors. Cancer Sci. 2025 Apr;116(4):1107-1114. doi: 10.1111/cas.16338. Epub 2025 Jan 29. PMID: 39888082
2024
- Yao Liang, Osamu Maeda, Chiaki Kondo, Kazuki Nishida, Yuichi Ando. Effects of KRAS, STK11, KEAP1, and TP53 mutations on the clinical outcomes of immune checkpoint inhibitors among patients with lung adenocarcinoma. PLoS One. 2024 Jul 22;19(7): e0307580. doi: 10.1371/journal.pone.0307580. eCollection 2024. PMID: 39037971 PMCID: PMC11262633
- Shigehiro Yagishita, Yuta Yamanaka, Takayasu Kurata, Kageaki Watanabe, Yukio Hosomi, Hidehito Horinouchi, Yuichiro Ohe, Yoshiro Nakahara, Katsuhiko Naoki, Tetsuhiko Asao, Kazuhisa Takahashi, Sho Saeki, Takuro Sakagami, Kazuhisa Nakashima, Yukari Tsubata, Yu Fujita, Hiroshi Wakui, Megumi Furuta, Jun Sakakibara Konishi, Mayu Ohuchi, Yuichi Ando, Hidenori Mizugaki, Akinobu Hamada. Multicenter pharmacokinetic and pharmaco-dynamic study of pembrolizumab for non-small cell lung cancer in patients aged 75 years and older. Clin Pharmacol Ther. 2024 Oct;116(4):1042-1051. doi: 10.1002/cpt.3339. Epub 2024 Jun 17. PMID: 38887810
- Osamu Maeda, Satoshi Furune, Mitsuro Kanda, Kazushi Miyata, Dai Shimizu, Shizuki Sugita, Kazuki Nishida, Masahiko Ando, Yasuhiro Kodera, Yuichi Ando. Docetaxel, cisplatin, and fluorouracil with pegfilgrastim on day 3 as neoadjuvant chemotherapy for esophageal cancer. Cancer Med. 2024 Jan;13(2):e6974. doi: 10.1002/cam4.6974. PMID: 38348961 PMCID: PMC10831909
- Yao Liang, Osamu Maeda, Kazushi Miyata, Mitsuro Kanda, Shizuki Sugita, Dai Shimizu, Kazuki Nishida, Yasuhiro Kodera, Yuichi Ando. Genetic polymorphisms as predictive biomarkers of adverse events during preoperative chemotherapy in esophageal cancer. Cancer Chemother Pharmacol. 2024 Feb;93(2):121-127. doi: 10.1007/s00280-023-04607-7 Epub 2023 Oct 28. PMID: 37898586
- Taigo Kato, Takahisa Kawaguchi, Taro Funakoshi, Yutaka Fujiwara, Yoshinari Yasuda, Yuichi Ando. Efficacy of Magnesium Supplementation in Cancer Patients Developing Hypomagnesemia Due to Anti-EGFR Antibody: A Systematic Review. Cancer Diagn Progn. 2024 Jul 3;4(4):390-395. doi: 10.21873/cdp.10337. eCollection 2024 Jul-Aug. PMID: 38962544 PMCID: PMC11215447
- Yao Liang, Osamu Maeda, Yuichi Ando. Biomarkers for immune-related adverse events in cancer patients treated with immune checkpoint inhibitors. Jpn J Clin Oncol Volume 54, Issue 4, Apr 2024, 365–375, https://doi.org/10.1093/jjco/hyad184. 05 Jan 2024 Article history. PMID: 38183211
- Taigo Kato, Shimon Kurasawa, Kentaro Takezawa, Yutaka Fujiwara, Yoshinari Yasuda, Yuichi Ando. Efficacy and safety of anti-angiogenic agents for cancer patients with proteinuria or a history of proteinuria: a systematic review. Anticancer Res. 2024 Mar;44(3):889-894. doi: 10.21873/anticanres.16882. PMID: 38423640
- Yao Liang, Yuichi Ando. UGT1A1 Testing for the Risk of Nanoliposomal Irinotecan-Related Toxicity. J Clin Oncol. 2024 Oct 28:JCO2401366. doi: 10.1200/JCO-24-01366. Online ahead of print. PMID: 39467220
- Yao Liang, Osamu Maeda, Tomoya Shimokata, Kenji Yokota, Hiroshi Koike, Tomohisa Sakai, Kunihiro Ikuta, Hiroshi Urakawa, Yoshihiro Nishida, Masashi Akiyama, Yuichi Ando. Potential drug interactions between pazopanib and proton pump inhibitors/potassium-competitive acid blockers in patients with soft tissue sarcoma. Int Cancer Conf J. 2023 Nov 16;13(1):63-67. doi: 10.1007/s13691-023-00638-2. eCollection 2024 Jan. PMID: 38187185
- Yuko Takano, Tomoya Shimokata, Hiroshi Urakawa, Toyone Kikumori, Yuichi Ando. Long-term response to MEK inhibitor monotherapy in a patient with papillary thyroid carcinoma harboring BRAF V600E mutation. Int Cancer Conf J. 2024 Apr 1;13(3):184-188. doi: 10.1007/s13691-024-00670-w. eCollection 2024 Jul. PMID: 38962055
2023
- Futoshi Ushijima, Tetsunari Hase, Yuki Yamashita, Hangsoo Kim, Tomoya Shimokata, Chiaki Kondo, Tomonori Sato, Tomoya Baba, Shohei Watanabe, Keisuke Futamura, Yuichi Ando, Masashi Mizuno, Makoto Ishii. Durvalumab plus carboplatin-etoposide treatment in a patient with small-cell lung cancer on hemodialysis: A case report and literature review. Int Cancer Conf J. 2023 Dec 21;13(2):88-92. doi: 10.1007/s13691-023-00640-8. eColle-ction 2024 Apr. PMID:38524642
- Yao Liang, Osamu Maeda, Kazushi Miyata, Mitsuro Kanda, Shizuki Sugita, Dai Shimizu, Kazuki Nishida, Yasuhiro Kodera, Yuichi Ando. Genetic polymorphisms as predictive biomarkers of adverse events during preoperative chemotherapy in esophageal cancer. Cancer Chemother Pharmacol. 2023 Oct 28. doi: 10.1007/s00280-023-04607-7. Online ahead of print. PMID: 37898586
- Yao Liang, Osamu Maeda, Kazushi Miyata, Mitsuro Kanda, Dai Shimizu, Shizuki Sugita, Tohru Okada, Junji Ito, Mariko Kawamura, Shunichi Ishihara, Nakatochi Masahiro, Masahiko Ando, Yasuhiro Kodera, Yuichi Ando. Feasibility assessment of global standard chemoradiotherapy followed by surgery in Japanese patients with esophageal cancer. Mol Clin Oncol. 2023 Mar 2;18(4):34.doi: 10.3892/mco.2023.2630. eCollection 2023 Apr. PMCID: PMC10011946
- Ayumu Matsuoka, Taichi Shimazu, Masahiro Takahashi, Fumio Nagashima, Hiroyuki Nishiyama, Maiko Fujimori, Yuichi Ando. A nationwide, cross-sectional, web-based survey on healthcare providers' knowledge about, attitudes toward, and perceived barriers to adherence to clinical practice guidelines for anticancer drug therapy for older patients with cancer in Japan. J Geriatr Oncol. 2023 Jan;14(1):101399. doi: 10.1016/j.jgo.2022.10.014. Epub 2022 Nov 7. PMID: 36357276
- Yuichi Ando, Hiroyuki Nishiyama, Hideki Shimodaira, Nao Takano, Emiko Sakaida, Koji Matsumoto, Koki Nakanishi, Hideki Sakai, Shokichi Tsukamoto, Keigo Komine, Yoshinari Yasuda, Taigo Kato, Yutaka Fujiwara, Takafumi Koyama, Hiroshi Kitamura, Takashige Kuwabara, Atsushi Yonezawa, Yuta Okumura, Kimikazu Yakushijin, Kazuki Nozawa, Hideaki Goto, Takeshi Matsubara, Junichi Hoshino, Motoko Yanagita. Chapter 3: Management of kidney injury caused by cancer drug therapy, from clinical practice guidelines for the management of kidney injury during anticancer drug therapy 2022. Int J Clin Oncol. 2023 Jul 16. doi: 10.1007/s10147-023-02382-2. Online ahead of print. PMID: 37453935
- Satoshi Furune, Yuichi Ando. Comment on: Carboplatin and irinotecan (CI) vs. Carboplatin and etoposide (CE) for the treatment of extended-stage small-cell lung cancer in an elderly population: A phase II/III randomized control trial. Lung Cancer. 2023 Dec:186:107416. doi: 10.1016/j.lungcan.2023.107416. Epub 2023 Oct 28. Online ahead of print. PMID: 37922770
- Yuichi Ando, Tomohiro Terada. Consent in pharmacogenetic testing. J Clin Oncol. 2023 Jun 2;JCO2300664. doi: 10.1200/JCO.23.00664. Online ahead of print. PMID: 37267582
2022
- Ayaka Ito, Kazuya Ichikawa, Masayuki Miyazaki, Atsunobu Sagara, Toshiki Motegi, Yuichi Ando, Koji Senzaki, Taku Nagai, Kiyofumi Yamada. Clinical impact of standardized creatinine on dose adjustment of capecitabine. Nagoya J Med Sci. 84(3) :547–553, 2022 Oct. doi:10.18999/nagjms.84.3.547. PMID: 36237888 PMCID: PMC9529624
- Osamu Maeda, Ayumu Matsuoka, Madoka Yanagawa, Yukie Muroyama, Kazuhisa Watanabe, Yao Liang, Takuya Ishikawa, Eizaburo Ohno, Hiroki Kawashima, Hiroyuki Umegaki, Masafumi Kuzuya, Yuichi Ando. Selection of chemotherapy for older patients with pancreatic cancer based on geriatric assessment. J Geriatr Oncol. 2022 Nov;13(8):1260-1263. doi: 10.1016/j.jgo.2022.09.005. Epub 2022 Sep 15. PMID: 36115797
- Ayako Mitsuma, Yasuki Ito, Tomoya Shimokata, Chie Tanaka, Kay Uehara, Goro Nakayama, Hiroko Terasaki, Yuichi Ando. Direct observation of retinal microvessels in cancer patients after systemic administration of bevacizumab and oxaliplatin. Cancer Diagn Progn. 2022 May 3;2(3):330-335. doi: 10.21873/cdp.10113. eCollection May-Jun 2022. PMID: 35530649 PMCID: PMC9066537
- Sachi Morita, Hironobu Minami, Ayako Mitsuma, Masanori Toyoda, Naomi Kiyota, Yuichi Ando. A phase I study of LCL161, a novel oral pan-inhibitor of apoptosis protein (IAP) antagonist, in Japanese patients with advanced solid tumors. Asia Pac J Clin Oncol. 2022 Oct;18(5):e427-e434. doi: 10.1111/ajco.13744. Epub 2022 Jan 30. PMID: 35098674. Online ahead of print.
- Osamu Maeda, Masahide Fukaya, Masahiko Koike, Kazushi Miyata, Mitsuro Kanda, Kazuki Nishida, Masahiko Ando, Yasuhiro Kodera, Yuichi Ando. Preoperative docetaxel, cisplatin, and fluorouracil treatment with pegfilgrastim on day 7 for patients with esophageal cancer: A phase II study. Asia Pac J Clin Oncol. 2022 Dec;18(6):578-585. doi: 10.1111/ajco.13755. Epub 2022 Jan 18. PMID: 35043574.
- Yuichi Ando, Sachi Morita, Tomoya Shimokata, Toyonori Tsuzuki, Shigeru Inafuku, Kenichiro Iwami, Nicoletta Brega, Takashi Akagawa, Toshiaki Tsujino, Tetsuya Ogawa. A rapid and durable response to larotrectinib in a patient with NTRK fusion-positive secretory carcinoma originating from the external auditory canal. Int Cancer Conf J. 2022 Jun 14;11(4):242-246.doi: 10.1007/s13691-022-00559-6. eCollection 2022 Oct. PMID: 36186229 PMCID: PMC9522948
2021
- Satoshi Nishiwaki, Yuichi Ando. COVID-19 pandemic and trends in clinical trials: a multi-region and global perspective. Front. Med. 2021 Dec 24;8:812370. doi: 10.3389/fmed. 2021.812370. eCollection 2021. PMID: 35004791 PMCID: PMC8739772
- Patrick Schöffski, Nicole Concin, Cristina Suarez, Vivek Subbiah, Yuichi Ando, Shiling Ruan, Joel P. Wagner, Keith Mansfield, Xu Zhu, Shizuka Origuchi, Sarah DiDominick, Carl U. Bialucha, Jason E. Faris, Ben Tran. A Phase 1 study of a CDH6-targeting antibody-drug conjugate in patients with advanced solid tumors with evaluation of inflammatory and neurological adverse events. Oncol Res Treat. 2021;44(10):547-556. doi: 10.1159/000518549. Epub 2021 Aug 19. PMID: 34515215
- Antje Wick, Oliver Bähr, Martin Schuler, Kristoffer Rohrberg, Sant Chawla, Janku Filip, David Schiff, Volker Heinemann, Yoshitaka Narita, Heinz-Josef Lenz, Masafumi Ikeda, Yuichi Ando, Wolfgang Wick, Joachim Steinbach, Michael Burger, Katharina Wenger, Ulrik Lassen, Kamalesh Sankhala, Cristiana Roggia, Isabelle Genvresse, Catya Munhoz, Christine Rentzsch, Susanne Reschke, Simon Langer, Markus Wagner, Stefan Kaulfuss, Charles Cai, Eleni Lagkadinou, Michael Jeffers, Carol Pena, Ghazaleh Tabatabai. Phase I Assessment of Safety and Therapeutic Activity of BAY1436032 in Patients with IDH1-Mutant Solid Tumors. Clin Cancer Res. 2021 May 15;27(10):2723-2733. doi: 10.1158/1078-0432.CCR-20-4256. Epub 2021 Feb 23. PMID: 33622704
- Hironobu Minami, Naomi Kiyota, Shiro Kimbara, Yuichi Ando, Tomoya Shimokata, Atsushi Ohtsu, Nozomu Fuse, Yasutoshi Kuboki, Toshio Shimizu, Noboru Yamamoto, Kazuto Nishio, Yutaka Kawakami, Shinichi Nihira, Kazuhiro Sase, Takahiro Nonaka, Hideaki Takahashi, Yukiko Komori, Koshin Kiyohara. Guidelines for clinical evaluation of anti-cancer drugs. Cancer Sci. 2021 Jul;112(7):2563-2577. doi: 10.1111/cas.14967. Epub 2021 Jun 8. PMID: 33990993
- Yuko Takano, Satoshi Furune, Yuki Miyai, Sachi Morita, Megumi Inoue, Tomoya Shimokata, Mihoko Sugishita, Ayako Mitsuma, Osamu Maeda, Yuichi Ando. Drug-induced thrombocytopenia associated with trastuzumab in a patient with HER2-positive recurrent gastric cancer. Int Cancer Conf J. 11, 67–70 2022. 02 Nov 2021 doi.org 10.1007/s13691-021-00520-z. PMID: 35116219 PMCID: PMC8786981
2020
- Takahiro Inaishi, Kenichi Fujita, Natsumi Matsumoto, Tomoya Shimokata, Osamu Maeda, Toyone Kikumori, Norifumi Hattori, Goro Nakayama, Yuichi Ando. Correlation between the metabolic conversion of a capecitabine metabolite, 5’-Deoxy-5-fluorocytidine,and creatinine clearance. In Vivo 34: 3539-3544 (2020) doi:10.21873/invivo.12196.
- Hironobu Minami, Yuichi Ando, Kenji Tamura, Randi Isaacs. Phase I study of LFA102 in patients with advanced breast cancer or castration-resistant prostate cancer. Anticancer Res 40: 5229-5235 (2020). doi:10.21873/anticanres.14526.
- Hironobu Minami, Toshihiko Doi, Masanori Toyoda, Yoshinori Imamura, Naomi Kiyota, Ayako Mitsuma, Tomoya Shimokata, Yoichi Naito, Nobuaki Matsubara, Takeshi Tajima, Kota Tokushige, Kae Ishihara, Scott Cameron, Yuichi Ando. Phase I dose-escalation study of the anti–PD-1 monoclonal antibody spartalizumab (PDR001) in Japanese patients with advanced malignancies. Cancer Sci. 2021;112:725–733. 2020 Oct 8. doi: 10.1111/cas. 14678.
- Akiko Ota, Sachi Morita, Ayumu Matsuoka, Tomoya Shimokata, Osamu Maeda, Ayako Mitsuma, Tetsuya Yagi, Takashi Asahara, Yuichi Ando.Detection of bacteria in blood circulation in patients receiving cancer chemotherapy.Int J Clin Oncol. 2020 Jan;25 (1):210-215. doi: 10.1007/s10147-019-01521-y. Epub 2019 Aug 12.
- Rie Tsuboi, Mihoko Sugishita, Yoshihisa Hirakawa, Yuichi Ando. Experiences and hidden needs of older patients, their families, and their physicians in palliative chemotherapy decision-making: a qualitative study. JJCO 2020 Apr 13;hyaa020. doi: 10.1093/jjco/Hyaa020.
2019
- Osamu Maeda, Ayumu Matsuoka, Kazuhiro Furukawa, Ryoji Miyahara, Yoshiki Hirooka and Yuichi Ando. Alterations in gene expression and DNA methylation profiles in gastric cancer cells obtained from ascitic fluids collected before and after chemotherapy. Mol Clin Oncol. 2019 Jul;11(1):91-98. doi:10.3892/mco.2019.1858. Epub 2019 May 10.
- Yuichi Ando, Satoru Iwasa, Shunji Takahashi, Hideo Saka, Tomoyuki Kakizume, Kazuto Natsume, Naoko Suenaga, Quadt Cornelia, Yasuhide Yamada. Phase I study of alpelisib (BYL719), an α-specific PI3K inhibitor, in Japanese patients with advanced solid tumors. Cancer Sci. 2019 Mar;110(3):1021-1031. doi: 10.1111/cas.13923. Epub 2019 Jan 30.
- Kan Yonemori, Makoto Kodaira, Taroh Satoh, Toshihiro Kudo, Shunji Takahashi, Kenji Nakano, Yuichi Ando, Tomoya Shimokata, Joji Mori, Koichi Inoue, Gerard Oakley, Sachi Sakaguchi, Kenji Tamura. A phase 1 study of olaratumab plus doxorubicin in Japanese patients with advanced soft-tissue sarcoma. Cancer Sci. 2018 Dec;109(12):3962-3970. doi: 10.1111/cas.13846
2018
- Bishal Gyawali, Tomoya Shimokata, Kazunori Honda, Yuichi Ando. Reporting harms more transparently in trials of cancer drugs. BMJ 2018; 363 doi: https://doi.org/10.1136/bmj.k4383 (Published 01 November 2018)
- Naomi Hayashi, Ayumu Matsuoka, Hidemi Goto, Momokazu Gotoh, Hitoshi Kiyoi, Yasuhiro Kodera, Masato Nagino, Fumio Nagashima, Yuichi Ando. Clinical effectiveness of geriatric assessment for predicting the tolerability of outpatient chemotherapy in older adults with cancer. J Geriatr Oncol. 2018 Jan;9(1):84-86. doi: 10.1016/j.jgo.2017.07.014. Epub 2017 Aug 9.
- Sachi Morita, Toyone Kikumori, Nobuyuki Tsunoda, Takahiro Inaishi, Yayoi Adachi, Akiko Ota, Masahiro Shibata, Ayumu Matsuoka, Kenichi Nakanishi, Dai Takeuchi, Takehumi Mizutani, Tomoya Shimokata, Hiromichi Hayashi, Osamu Maeda and Yuichi Ando. Feasibility of dose-dense epirubicin and cyclophosphamide with subcutaneous pegfilgrastim 3.6 mg support: a single-center prospective study in Japan. Int J Clin Oncol. 2018 Feb;23(1):195-200. doi: 10.1007/s10147-017-1177-z. Epub 2017 Aug 8.
- Horie Shigeo, Mototsugu Oya, Masaomi Nangaku, Yoshinari Yasuda, Yasuhiro Komatsu, Motoko Yanagita, Yuko Kitagawa, Hiroyuki Kuwano, Hiroyuki Nishiyama, Chikashi Ishioka, Hiromasa Takaishi, Hideki Shimodaira, Akira Mogi, Yuichi Ando, Koji Matsumoto, Daisuke Kadowaki, Satoru Muto. Guidelines for treatment of renal injury during cancer chemotherapy 2016. Clin Exp Nephrol. 2018 Feb;22(1):210-244. doi: 10.1007/s10157-017-1448-z.
2017
- Hiroaki Tsukuura, Masayuki Miyazaki, Tatsuya Morita, Mihoko Sugishita, Hiroshi Kato, Yuka Murasaki, Bishal Gyawali, Yoko Kubo, Masahiko Ando, Masashi Kondo, Kiyofumi Yamada, Yoshinori Hasegawa and Yuichi Ando. Efficacy of Prophylactic Treatment for Oxycodone-Induced Nausea and Vomiting Among Patients with Cancer Pain (POINT): A Randomized, Placebo-Controlled, Double-Blind Trial.2018 Mar;23(3):367-374. doi: 10.1634/theoncologist.2017-0225. Epub 2017 Oct 16.
- Osamu Maeda, Ayumu Matsuoka, Ryoji Miyahara, Kohei Funasaka, Yoshiki Hirooka, Masahide Fukaya, Masato Nagino, Yasuhiro Kodera, Hidemi Goto and Yuichi Ando. Modified docetaxel, cisplatin and capecitabine for stage IV gastric cancer in Japanese patients: A feasibility study. World J Gastroenterol. 2017 Feb 14;23(6):1090-1097. doi: 10.3748/wjg.v23.i6.1090.
- Bishal Gyawali, Tomoya Shimokata, Masahiko Ando, Kazunori Honda and Yuichi Ando. Risk of serious adverse events and fatal adverse events with sorafenib in patients with solid cancer: a meta-analysis of phase 3 randomized controlled trials. Ann Oncol. 2017 Feb 1;28(2):246-253. doi: 10.1093/annonc/mdw549.
2016
- Ayumu Matsuoka, Osamu Maeda, Takefumi Mizutani, Yasuyuki Nakano, Nobuyuki Tsunoda, Toyone Kikumori, Hidemi Goto, Yuichi Ando. Bevacizumab exacerbates paclitaxel-induced neuropathy: a retrospective cohort study. PLoS One. 2016 Dec 19;11(12):e0168707. doi: 10.1371/journal.pone.0168707.
- Bishal Gyawali, Tomoya Shimokata, Masahiko Ando, Kazunori Honda and Yuichi Ando. Risk of serious adverse events and fatal adverse events with sorafenib in patients with solid cancer: a meta-analysis of phase 3 randomized controlled trials. Ann Oncol. 2016 Oct 22. pii: mdw549.
- Hironobu Minami, Yuichi Ando, Brigette Buig Yue Ma, Jih-Hsiabg Lee, Hiroyuki Momota, Yutaka Fujiwara, Leung Li, Koichi Fukino, Koji Ito, Takeshi Tajima, Asuka Mori and Chia-Chi Lin. A phase 1, multicenter, open-label, dose-escalation study of sonidegib in Asian patients with advanced solid tumors. Cancer Sci. 2016 Oct;107(10):1477-1483. doi: 10.1111/cas.13022.
- Bishal Gyawali, Tomoya Shimokata, Kazunori Honda, Chihiro Kondoh, Naomi Hayashi, Yasushi Yoshino, Naoto Sassa, Yasuyuki Nakano, Momokazu Gotoh, and Yuichi Ando. Long term use of mTOR inhibitors leads to loss of muscle mass. Mol Clin Oncol. 2016 Nov;5(5):641-646.
- Ayumu Matsuoka, Ayako Mitsuma, Osamu Maeda, Hiroaki Kajiyama, Hitoshi Kiyoi, Yasuhiro Kodera, Masato Nagino, Hidemi Goto, Yuichi Ando. Quantitative assessment of chemotherapy-induced peripheral neurotoxicity using a point-of-care nerve conduction device. Cancer Sci. 2016 Oct;107(10):1453-1457. doi: 10.1111/cas.13010.
- Goro Nakayama, Tsutomu Fujii, Kenta Murotani, Keisuke Uehara, Norifumi Hattori, Masamichi Hayashi, Chie Tanaka, Daisuke Kobayashi, Mitsuro Kanda, Suguru Yamada, Hiroyuki Sugimoto, Masahiko Koike, Michitaka Fujiwara, Yuichi Ando and Yasuhiro Kodera. Modified two-dimensional response as surrogate marker of overall survival in patients with metastatic colorectal cancer. Cancer Sci. 2016 Oct;107(10):1492-1498. doi: 10.1111/cas.13023.
- Kazunori Honda, Kyosuke Takeshita, Kenta Murotani, Ayako Mitsuma, Hironori Hayashi, Nobuyuki Tsunoda, Toyone Kikumori, Toyoaki Murohara, Yuichi Ando. Assessment of left ventricular diastolic function during trastuzumab treatment in patients with HER2-positive breast cancer. Breast Cancer. 2017 Mar;24(2):312-318. doi: 10.1007/s12282-016-0705-4.
- Koichiro Watanabe, Satoshi Otsu, Yoshinori Hirashima, Ryotaro Morinaga, Kazuo Nishikawa, Yasushi Hisamatsu, Tomoya Shimokata, Megumi Inada-Inoue, Takashi Shibata, Hiromi Takeuchi, Takahiro Watanabe, Kota Tokushige, Heiko Maacke, Kuniaki Shirao, Yuichi Ando. A phase I study of binimetinib (MEK162) in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol. 2016 Jun;77(6):1157-64. doi: 10.1007/s00280-016-3019-5. Epub 2016 Apr 12.
- Masashi Takano, Kaichiro Yamamoto, Tsutomu Tabata, Yuji Minegishi, Takuma Yokoyama, Eiji Hirata, Takeshi Ikeda, Muneaki Shimada, Kouzo Yamada, Satoshi Morita, Yuichi Ando, Yukihiro Okutani, Masahiro Sugihara, Toru Sugiyama, Yasuo Ohashi, and Yuh Sakata. The impact of UGT1A1 genotype upon toxicities of combination with low-dose irinotecan plus platinum. Asia Pac J Clin Oncol. 2016 Jun;12(2):115-24. doi: 10.1111/ajco.12453. Epub 2016 Feb 10.
- Mihoko Sugishita, Tsuneo Imai, Toyone Kikumori, Ayako Mitsuma, Tomoya Shimokata, Takashi Shibata, Sachi Morita, Megumi Inada-Inoue, Masataka Sawaki, Yoshinori Hasegawa and Yuichi Ando. Pharmacogenetic association between GSTP1 genetic polymorphism and febrile neutropenia in Japanese patients with early breast cancer. Breast Cancer 23:195–201, 2016
- Bishal Gyawali, Tomoya Shimokata, Kazunori Honda, Hiroaki Tsukuura, Yuichi Ando. Should low-income countries invest in breast cancer screening? Cancer Causes Control (2016). doi:10.1007/s10552-016-0812-8 in press
- Bishal Gyawali, Tomoya Shimokata, Kazunori Honda and Yuichi Ando. Chemotherapy in locally advanced head and neck squamous cell carcinoma. Cancer Treat Rev. 2016 Mar;44:10-6. doi: 10.1016/j.ctrv.2016.01.002. Epub 2016 Jan 22.
- Ota A, Gyawali B, Matsuoka A, Ando Y. Unconvincing benefit of combination therapy with gefitinib and pemetrexed in advanced non-small-cell lung cancer. J Clin Oncol. 2017 Feb 20;35(6):691-692. doi: 10.1200/JCO.2016.70.4148. Epub 2016 Dec 5.
- Bishal Gyawali, Yuichi Ando. Adjuvant Sunitinib for high-risk resected renal cell carcinoma: a meta-analysis of ASSURE and S-TRAC trials. Ann Oncol (2016) mdw667. DOI: https://doi.org/10.1093/annonc/mdw667 Published: 19 December 2016
- Bishal Gyawali, Akiko Ota, Yuichi Ando. Explanation for the shorter progression-free survival with Nivolumab in Checkmate 057. N Engl J Med. 2016 Feb 4;374(5):493.
- Eiko Yamamoto, Kaoru Niimi, Kayo Fujikake, Tetsuya Nishida, Makoto Murata, Ayako Mitsuma, Yuichi Ando and Fumitaka Kikkawa. High‑dose chemotherapy with autologous peripheral blood stem cell transplantation for choriocarcinoma: A case report and literature review. Mol Clin Oncol. 2016 Nov;5(5):660-664. Epub 2016 Sep 8.
- Osamu Maeda, Kenji Yokota, Naoki Atsuta, Masahisa Katsuno, Masashi Akiyama, Yuichi Ando. Nivolumab for the treatment of malignant melanoma in a patient with pre-existing myasthenia gravis. Nagoya J Med Sci. 2016 Feb;78(1):119-22.
2015
- Takashi Shibata, Tomoki Ebata, Ken-ichi Fujita, Tomoya Shimokata, Osamu Maeda, Ayako Mitsuma, Yasutsuna Sasaki, Masato Nagino and Yuichi Ando. Optimal dose of gemcitabine for the treatment of biliary tract or pancreatic cancer in patients with liver dysfunction. Cancer Sci. 2016 Feb;107(2):168-72. doi: 10.1111/cas.12851. Epub 2016 Feb 2.
- Naomi Hayashi, Yuichi Ando, Bishal Gyawali, Tomoya Shimokata, Osamu Maeda, Masahide Fukaya, Hidemi Goto, Masato Nagino and Yasuhiro Kodera. Low skeletal muscle density is associated with poor survival in patients with metastatic gastric cancer receiving chemotherapy. Oncol Rep. 2016 Mar;35(3):1727-31. doi: 10.3892/or.2015.4475. Epub 2015 Dec 8.
- Ayumu Matsuoka, Osamu Maeda, Megumi Inada-Inoue, Eizaburo Ohno, Yoshiki Hirooka, Yukihiro Yokoyama, Tsutomu Fujii, Masato Nagino, Hidemi Goto and Yuichi Ando. FOLFIRINOX-induced reversible dysarthria. Oncol Lett. 2015 Oct;10(4):2662-2664. Epub 2015 Aug 11.
- Naomi Kiyota, Martin Schlumberger, Kei Muro, Yuichi Ando, Shunji Takahashi, Yasukazu Kawai, Lori Wirth, Bruce Robinson, Steven Sherman, Takuya Suzuki, Katsuki Fujino, Anubha Gupta, Seiichi Hayato and Makoto Tahara. A subgroup analysis of Japanese patients in a phase 3 study of lenvatinib in radioiodine-refractory differentiated thyroid cancer. Cancer Sci. 2015 Dec;106(12):1714-21. doi: 10.1111/cas.12826. Epub 2015 Nov 2.
- Hiroaki Tsukuura, Yuichi Ando, Bishal Gyawali, Masami Matsumoto, Mihoko Sugishita, Kazunori Honda, Hiroshi Urakawa, Osamu Maeda, and Yoshinori Hasegawa. Prophylactic use of antiemetics for prevention of opioidinduced nausea and vomiting: a questionnaire survey among Japanese physicians. J Palliat Med. 2015;18:977-80. doi: 10.1089/jpm.2015.0203.
- Hirofumi Mukai, Norikazu Masuda, Hiroshi Ishiguro, Ayako Mitsuma, Takashi Shibata, Jun Yamamura, Masakazu Toi, Aiko Watabe, Akiko Sarashina, Martina Uttenreuther-Fischer, Yuichi Ando. Phase I trial of afatinib plus vinorelbine in Japanese patients with advanced solid tumors, including breast cancer. Cancer Chemother Pharmacol 2015 Oct;76(4):739-50.
- Francis Worden, Martin Fassnacht, Yuankai Shi, Tatiana Hadjieva, Françoise Bonichon, Ming Gao, Laura Fugazzola, Yuichi Ando, Yasuhisa Hasegawa, Do Joon Park, Young Kee Shong, Johannes W.A. Smit, John Chung, Christian Kappeler, Gerold Meinhardt, Martin Schlumberger and Marcia S. Brose. Safety and tolerability of sorafenib in patients with radioiodine-refractory thyroid cancer. Endocr Relat Cancer. 2015 Dec;22(6):877-87. doi: 10.1530/ERC-15-0252.
- Kanako Shibata, Yoshinari Yasuda, Ryo Kobayashi, Yuichi Ando, Tomoya Shimokata, Hideki Kamiya, Mutsuharu Hayashi, Shoichi Maruyama, Seiichi Matsui, Makoto Nakao, Teruo Tsuchiya and Hitomi Teramachi. Renal function evaluation in patients with cancer who were scheduled to receive carboplatin or S-1. Clin Exp Nephrol. 2015 Dec;19(6):1107-13. doi: 10.1007/s10157-015-1115-1. Epub 2015 Apr 19.
- Yoshihiro Nishida, Satoshi Tsukushi, Hiroshi Urakawa, Shunsuke Hamada, Eiji Kozawa, Kunihiro Ikuta, Yuichi Ando, Naoki Ishiguro. Low-dose chemotherapy with methotrexate and vinblastine for patients with desmoid tumors: relationship to CTNNB1 mutational status. Int J Clin Oncol 20:1211–1217, 2015
- Wataru Ichikawa, Keisuke Uehara, Keisuke Minamimura, Chihiro Tanaka, Yasumasa Takii, Hideaki Miyauchi, Sotaro Sadahiro, Ken-ichi Fujita, Toshikazu Moriwaki, Masato Nakamura, Takehiro Takahashi, Akihito Tsuji, Katsunori Shinozaki, Satoshi Morita, Yuichi Ando, Yukihiro Okutani, Masahiro Sugihara, Toru Sugiyama, Yasuo Ohashi, Yuh Sakata. An internally and externally validated nomogram for predicting the risk of irinotecan-induced severe neutropenia in advanced colorectal cancer patients. Br J Cancer. 2015 May 12;112(10):1709-16. doi: 10.1038/bjc.2015.122. Epub 2015 Apr 16.
- Tomoyo Oguri, Tomoya Shimokata, Isao Ito, Yoshinari Yasuda, Naoto Sassa, Masami Nishiyama, Akinobu Hamada, Yoshinori Hasegawa, and Yuichi Ando. Extension of the Calvert formula to patients with severe renal insufficiency. Cancer Chemother Pharmacol. 2015 Jul;76(1):53-9. doi: 10.1007/s00280-015-2769-9.
- Masayuki Umemura, Akio Ito, Yuichi Ando, Kiyofumi Yamada, Yoshifumi Wakiya and Toshitaka Nabeshima. Effects of outside air temperature on the preparation of antineoplastic drug solutions in biological safety cabinets. J Oncol Pharm Pract. Aug;21(4):243-8, 2015
- Bishal Gyawali, Naomi Hayashi, Hiroaki Tsukuura, Kazunori Honda, Tomoya Shimokata, Yuichi Ando. Opioid-induced constipation. Scand J Gastroenterol. Scandinavian Journal of Gastroenterology 2015 Nov;50(11):1331-8. doi: 10.3109/00365521.2015.1054423. Epub 2015 Jun 10.
- Ayumu Matsuoka, Osamu Maeda and Yuichi Ando. TRIBE study: administration of all three cytotoxic agents is crucial in advanced colorectal cancer. Lancet Oncol. 2015 Dec;16(16):e577. doi: 10.1016/S1470-2045(15)00390-3.
- Kazunori Honda, Bishal Gyawali and Yuichi Ando. Reasons for discrepancy between locoregional and distant PFS in the trial of locoregional treatment for metastatic breast cancer. Lancet Oncol. 2015 Dec;16(16):e586. doi: 10.1016/S1470-2045(15)00368-X
- Bishal Gyawali, Tomoya Shimokata and Yuichi Ando. Discordance between the results and conclusions of ICON-7. Lancet Oncol. 2015 Oct;16(13):e478. doi: 10.1016/S1470-2045(15)00283-1.
- Bishal Gyawali, Tomoya Shimokata, Kazunori Honda and Yuichi Ando. Flaws in the trial design of IFCT-0802. Ann Oncol. 2015 Jun 25. pii: mdv273.
- Sachi Morita, Mariko Hiramatsu, Mihoko Sugishita, Bishal Gyawali, Takashi Shibata, Tomoya Shimokata, Hiroshi Urakawa, Ayako Mitsuma, Suzuko Moritani, Toshinobu Kubota, Shu Ichihara and Yuichi Ando. Pazopanib monotherapy in a patient with malignant granular cell tumor that originally arose in the right orbit: a case report. Oncol Lett. 2015 Aug;10(2):972-974. Epub 2015 May 26.
2014
- Toru Nakanishi, Daisuke Aoki, Yoh Watanabe, Yuichi Ando, Naoki Tomotsugu, Yuji Sato, and Toshiaki Saito. A phase II clinical trial of pegylated liposomal doxorubicin and carboplatin in Japanese patients with platinum-sensitive recurrent ovarian, fallopian tube, or primary peritoneal Cancer. Jpn J Clin Oncol. 2015 May;45(5):422-6. doi: 10.1093/jjco/hyv016. Epub 2015 Feb 10.
- Maeda O, Ando T, Ishiguro K, Watanabe O, Miyahara R, Nakamura M, Funasaka K, Kazuhiro F, Ando Y, Goto H. Safety of repeated cell-free and concentrated ascites reinfusion therapy for malignant ascites from gastrointestinal cancer. Mol Clin Oncol. 2014 Nov;2(6):1103-1106. Epub 2014 Jul 4.
- Megumi Inada-Inoue, Yuichi Ando, Kenji Kawada, Ayako Mitsuma, Masataka Sawaki, Taro Yokoyama, Yu Sunakawa, Hiroo Ishida, Kazuhiro Araki, Keishi Yamashita, Keiko Mizuno, Fumio Nagashima, Akiko Takekura, Kazuo Nagamatsu, Yasutsuna Sasaki. Phase 1 study of pazopanib alone or combined with lapatinib in Japanese patients with solid tumors. Cancer Chemother Pharmacol 73:673-83, 2014
- Goro Nakayama, Chie Tanaka, Keisuke Uehara, Kiyoshi Ishigure, Hiroyuki Yokoyama, Daisuke Kobayashi, Mituro Kanda, Suguru Yamada, Tsutomu Fujii, Hiroyuki Sugimoto, Masahiro Koike, Shuji Nomoto, Michitaka Fujiwara, Yuichi Ando and Yasuhiro Kodera. The impact of dose/time modification in irinotecan and oxaliplatin based chemotherapies on outcomes in colorectal cancer. Cancer Chemother Pharmacol 2014 Apr;73(4):847-55. doi: 10.1007/s00280-014-2416-x. Epub 2014 Feb 28.
- Yuichi Ando, Megumi Inada-inoue, Ayako Mitsuma, Takayuki Yoshino, Atsushi Ohtsu, Naoko Suenaga, Masahiko Sato, Tomoyuki Kakizume, Matthew Robson, Cornelia Quadt and Toshihiko Doi. A Phase I dose-escalation study of buparlisib (BKM120), an oral pan-class I PI3K inhibitor, in Japanese patients with advanced solid tumors. Cancer Sci 105(3):347-53, 2014
- Yuichiro Yoshioka, Keisuke Uehara, Tomoki Ebata, Yukihiro Yokoyama, Ayako Mitsuma, Yuichi Ando and Masato Nagino. Postoperatibe complications following neoadjuvant bevacizumab treatment for for advanced colorectal cancer. Surg Today. 2014 Jul;44(7):1300-6. doi: 10.1007/s00595-013-0686-2. Epub 2013 Aug 14.
- Takashi Shibata, Yosuke Minami, Ayako Mitsuma, Sachi Morita, Megumi Inada-Inoue, Tomoyo Oguri, Tomoya Shimokata, Mihoko Sugishita, Tomoki Naoe and Yuichi Ando. Association between severe toxicity of nilotinib and UGT1A1 polymorphisms in Japanese patients with chronic myelogenous leukemia. Int J Clin Oncol 19(2);391-396, 2014
- Yaping Shou, Douglas Robinson, Dereck Amakye, Kristine Rose, Yoon-Jae Cho, Keith L. Ligon, Thad Sharp, Asifa Haider, Raj Bandaru, Yuichi Ando, Birgit Geoerger, Francois Doz, David Ashley, Darren Hargrave, Michela Casanova, Jordi Rodon, Anne Thomas, Alain Mita, Tobey MacDonald, Mark Kieran. Five-gene hedgehog signature developed as a patient preselection tool for hedgehog inhibitor therapy in medulloblastoma. Clin Cancer Res. 2015 Feb 1;21(3):585-93. doi: 10.1158/1078-0432.CCR-13-1711. Epub 2014 Dec 3.
- Yuichi Ando, Tomoya Shimokata, Yoshinari Yasuda, and Yoshinori Hasegawa. Carboplatin dosing for adult Japanese patients (invited review). Nagoya J Med Sci 76: 1-9, 2014
- Ayumu Matsuoka, Osamu Maeda and Yuichi Ando. FOLFOXIRI and bevacizumab for metastatic colorectal cancer. Letter to the editor regarding the Loupakis article of 23-Oct-2014 [correspondence]. N Engl J Med; 372:290-292, 2015
- Bishal Gyawali, Hiroaki Tsukuura, Kazunori Honda, Tomoya Shimokata and Yuichi Ando. Some questions on the randomized controlled trial of communication skills training for oncologists. J Clin Oncol 2015 Jan 10;33(2):222. doi: 10.1200/JCO.2014.58.4532. Epub 2014 Dec 1.
- Bishal Gyawali, Bishesh Poudyal, Tomoya Shimokata and Yuichi Ando. Cancer care and research in India: what does it mean to Nepal?\\\" Lancet Oncol. 2014 Jul;15(8):e299-300. doi: 10.1016/S1470-2045(14)70230-X.
- Tomoya Shimokata and Yuichi Ando. Carboplatin plus paclitaxel scheduling for advanced ovarian cancer. Lancet Oncol. 2014 Jun;15(7):e249-50. doi: 10.1016/S1470-2045(14)70214-1
- Yuji Miura, Chiyo K Imamura, Koya Fukunaga, Yoshihiko Katsuyama, Koichi Suyama, Toshikazu Okaneya, Taisei Mushiroda, Yuichi Ando, Toshimi Takano and Yusuke Tanigawara. Sunitinib-induced severe toxicities in a Japanese patient with the ABCG2 421 AA genotype. BMC Cancer. 2014 Dec 16;14:964. doi: 10.1186/1471-2407-14-964.
- Yasuhito Funahashi, Naoto Sassa, Megumi Inada-Inoue, Yuichi Ando, Yoshihisa Matsukawa and Momokazu Gotoh. Acute aortic dissection in a patient receiving multiple tyrosine kinase inhibitors for 5 Years. Aktuelle Urol. 45(2):132-134, 2014
- Osamu Maeda, Takafumi Ando, Kazuhiro Ishiguro, Osamu Watanabe, Ryoji Miyahara, Masanao Nakamura, Kohei Funasaka, Kazuhiro Furukawa, Yuichi Ando, Katsuhiko Kato and Hidemi Goto. A patient with esophageal cancer with bone metastasis who achieved pain relief with repetitive administration of strontium-89 chlorid. Clin J Gastroenterol. 2014 Oct;7(5):387-91.
- Osamu Maeda, Takafumi Ando, Kazuhiro Ishiguro, Osamu Watanabe, Ryoji Miyahara, Tomoko Miyata, Keita Itatsu, Yuichi Ando and Hidemi Goto. A case of gastric carcinosarcoma with distant metastasis for which chemotherapy with S-1 plus cisplatin was transiently effective. Int Canc Conf J (2014) 3:252–259
- Yuichi Ando. Cytochrome P450 and anticancer drugs. In. Handbook of Anticancer Pharmacokinetics and Pharmacodynamics (Cancer Drug Discovery and Development) Michelle A. Rudek, Cindy H. Chau, William Figg, Howard L. McLeod, eds. pp. 273-288. Springer; 2nd ed. 2014 (2014/1/31) ,
MESSAGE
Call for graduate students
The curriculum outlines the broad concepts, related learning objectives and the associated theoretical knowledge, clinical skills, attitudes and behaviors required and commonly utilized by medical oncology physicians within and outside Japan. At the completion of the PhD in the program, students should be competent to provide at consultant level, unsupervised comprehensive medical care in medical oncology.
The training includes aspects of basic science and internal medicine course relevant to oncology together with basics and advanced training of medical oncology and chemotherapy. The curriculum requires a professional approach from the students who will be expected to have a deep understanding of the subjects. It is expected that the trainees will read the texts and make critical use, where appropriate of original literature and peer scrutinized review articles in the related scientific and clinical literature such that they can aspire to an excellent standard in the practice of medical oncology. Learning methods include interdisciplinary meetings, hospital rounds and outpatient departments with medical oncologists, seminars and workshops, journal club, textbooks, and clerkships.
Especially, this course aims to learn evidence-based cancer chemotherapy and palliative medicine, together with clinical pharmacology, clinical trials, clinical safety, and medical ethics. Students will learn clinical pharmacology and pharmacogenetics of anticancer agents by investigating interindividual variations in drug response and toxicity to cancer chemotherapy and by conducting some clinical trials of cancer chemotherapy.

