Cell Physiology
KEYWORDS
- Neuron
- Synapse
- Plasticity
- Patch clamp
- Auditory
- Oligodendrocyte
- Axon initial segment
- Imaging
- Genome editing
HEAD

LAB MEMBER
| Faculty | Position | Researchers |
|---|---|---|
| SAKAMOTO Hirokazu | Associate Professor | Researchers |
| EGAWA Ryo | Assistant Professor | Researchers |
| FUKAYA Ryota | Assistant Professor | Researchers |
| ADACHI Ryota | Assistant Professor | Researchers |
CONTACT
| kuba◎med.nagoya-u.ac.jp(Please send a message after replacing "◎" mark with "@" mark. ) | |
| HP | Private Page |
OUTLINE
Our lab aims to understand fundamental principles on formation and operation of neural circuits in the brain, particularly focusing on experience-dependent reorganizations of neuron and synapse in the auditory system.
RESEARCH PROJECTS
Brain is composed of over 100 billions of neurons. Neurons are highly differentiated in their morphological and biophysical properties. Furthermore, they are interconnected with each other via synapses, thus creating complex neural circuits. Recent advances in neuroscience give us a wealth of knowledge on the anatomical basis of neural circuits and also the properties of individual neurons and synapses. However, how these features actually shape specific functions of neural circuits is not well understood. We are addressing this issue by multidisciplinary approaches, including electrophysiology, optics, morphology, molecular biology, and computations.
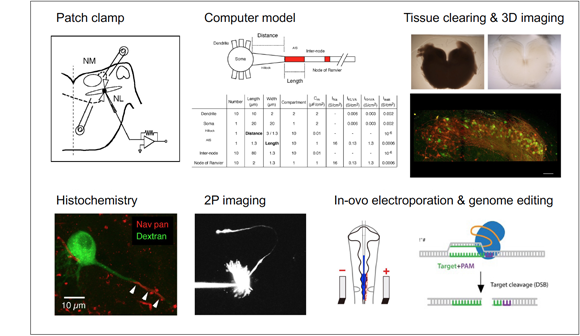
Cellular and molecular mechanisms of binaural spatial hearing
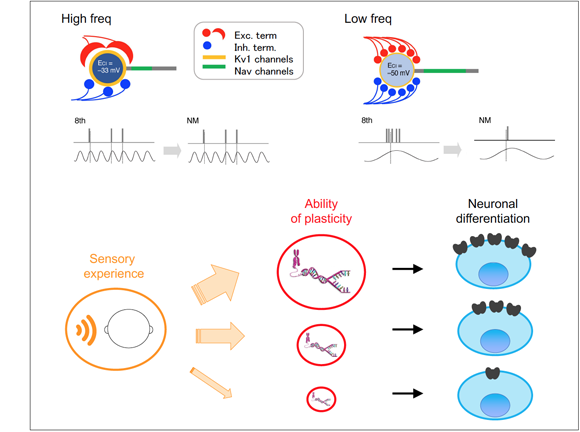
Sound localization is a behavior to identify the direction of sounds and requires detecting a difference in sound arrival times between the two ears. We can resolve as small as 1 degree of sound source changes along the horizontal plane, which corresponds to 10 microseconds of interaural time difference. This resolution of time is outstanding, considering that action potentials often have duration in the order of a millisecond in the brain. We are interested in how such a small time difference is extracted in brainstem auditory circuits, particularly focusing on tonotopic differentiation of neurons within the circuits. We are also studying how the properties of these circuits are acquired and mature in response to sensory inputs during development.
Structural plasticity at the axon initial segment
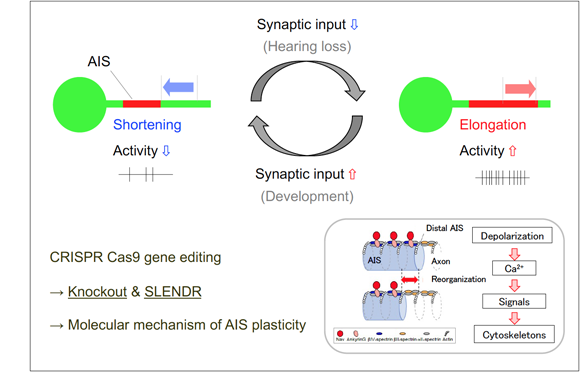
Axon initial segment (AIS) is a highly specialized neuronal structure that separates axonal and somatodendritic compartments. AIS is enriched with voltage-gated Na channels, and plays a critical role in initiating action potentials. We showed in brainstem auditory neurons that deprivation auditory nerve activity increased the length of AIS, thereby enhancing the excitability of the neurons. This indicates that AIS is highly plastic and changes its structure to regulate neuronal activity. Neural activity is crucial for the maintenance of neural circuit. Thus, this reorganization of AIS may work as a homeostatic mechanism to compensate for the loss of auditory nerve activity, thereby contributing to the maintenance of auditory circuits after hearing loss. We are now elucidating properties, mechanisms, and function of this AIS plasticity. The findings will reinforce our knowledge on how neural circuit function is achieved and maintained in an experience dependent manner.
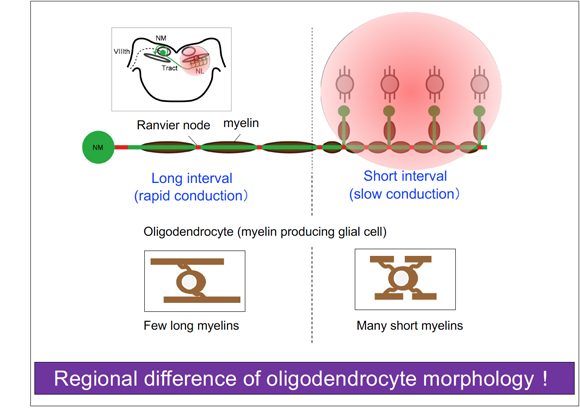
Mechanisms underlying region-specific spacing of Ranvier nodes
We are studying the mechanisms by which oligodendrocytes regulate conduction velocity along axons in a region specific manner.
BIBLIOGRAPHY
2025
- Sato R, Adachi R, Yokoi N, Tsujimura T, Egawa R, Hara Y, Fukata Y, Fukata M, Ogi T, Sone M, *Kuba H Loss of neuronal activity facilitates surface accumulation of p75NTR and cell death in avian cochlear nucleus. Neurosci. Res. 213, 23-34 (2025).
- *Egawa R, Yawo H, Kuba H Activity-dependent refinement of axonal projections forms one-to-one connection pattern in the developing chick ciliary ganglion. Front. Cell. Neurosci. 9:1560402 (2025).
- Egawa R, Hiraga K, Matsui R, Watanabe D, *Kuba H Regional heterogeneities of oligodendrocytes underlie biased Ranvier node spacing along single axons in sound localization circuit. eLife 14:RP106415(2025).
2023
- Jahan I, Adachi R, Egawa R, Nomura H, *Kuba H CDK5/p35-dependent microtubule reorganization contributes to homeostatic shortening of the axon initial segment. J. Neurosci. 43:359-372 (2023).
2022
- *Yamada R, *Kuba H Cellular strategies for frequency-dependent computation of interaural time difference. Front. Synaptic. Neurosci 14:891740 (2022).
2021
- Al-Yaari M, Onogi C, Yamada R, Adachi R, Kondo D, Kuba H. Tonotopic specializations in number, size, and reversal potential of GABAergic inputs fine-tune temporal coding at avian cochlear nucleus. J. Neurosci. 2021; 41:8904-8916
- Yamada R, Kuba H. Dendritic synapse geometry optimizes binaural computation in a sound localization circuit. Sci. Adv. 2021; 7:eabh0024
2020
- Al-Yaari M, Yamada R, Kuba H. Excitatory-inhibitory synaptic coupling in avian nucleus magnocellularis. J. Neurosci. 2020; 40:619-631.
- Akter N, Fukaya R, Adachi R, Kawabe H, Kuba H. Structural and functional refinement of the axon initial segment in avian cochlear nucleus during development. J. Neurosci. 2020; 40:6709-6721.
2019
- Adachi R, Yamada R, Kuba H. Tonotopic Differentiation of Coupling between Ca2+ and Kv1.1 Expression in Brainstem Auditory Circuit. iScience 2019; 13:199-213.
2018
- Fukaya R, Yamada R, *Kuba H. Tonotopic Variation of the T-Type Ca2+ Current in Avian Auditory Coincidence Detector Neurons. J. Neurosci. 2018; 38:335-346.
- Akter N, Adachi R, Kato A, Fukaya R, Kuba H. Auditory input shapes tonotopic differentiation of Kv1.1 expression in avian cochlear nucleus during late development. J. Neurosci. 2018; 38:2967-2980.
2016
- Yamada R, Kuba H. Structural and Functional Plasticity at the Axon Initial Segment. Front. Cell Neurosci. 2016; 10:250.
- Susuki K, Kuba H. Activity-dependent regulation of excitable axonal domains. J. Physiol. Sci. 2016; 66, 99-104.
2015
- Kuba H, Yamada R, Ishiguro G, Adachi R. Redistribution of Kv1 and Kv7 enhances neuronal excitability during structural axon initial segment plasticity. Nat. Commun. 2015; 6:8815.
- Adachi R, Yamada R, Kuba H. Plasticity of the axonal trigger zone. Neuroscientist, 2015; 21: 255-265.
2014
- Kuba H, Adachi R, Ohmori H. Activity-dependent and activity-independent development of the axon initial segment. J Neurosci, 2014; 34: 3433-3453.
2013
- Yamada R, Okuda H, Kuba H, Nishino E, Ishii TM, Ohmori H. The cooperation of sustained and phasic inhibitions increases the contrast of ITD-tuning in low-frequency neurons of the chick nucleus laminaris. J Neurosci, 2013; 33: 3927-3938.
- Okuda H, Yamada R, Kuba H, Ohmori H. Activation of metabotropic glutamate receptors improves the accuracy of coincidence detection by presynaptic mechanisms in the nucleus laminaris of the chick. J Physiol (Lond), 2013; 591: 365-378.
2012
- Kuba H. Structural tuning and plasticity at the axon initial segment in auditory neurons. J Physiol (Lond), 2012; 590: 5571-5579.
2011
- Grubb MS, Shu Y, Kuba H, Rasband MN, Wimmer VC, Bender KJ. Short- and long-term plasticity at the axon initial segment. J Neurosci, 2011; 31: 16045-16055.
2010
- Kuba H, Oichi Y, Ohmori H. Presynaptic activity regulates Na+ channel distribution at the axon initial segment. Nature, 2010; 465: 1075-1078.
2009
- Kuba H, Ohmori H. Roles of axonal sodium channels in precise auditory time coding at nucleus magnocellularis of the chick. J Physiol (Lond), 2009; 587: 87-100.
2006
- Kuba H, Ishii TM, Ohmori H. Axonal site of spike initiation enhances auditory coincidence detection. Nature, 2006; 444: 1069-1072.
2005
- Yamada R, Kuba H, Ishii TM, Ohmori H. Hyperpolarization-activated cyclic nucleotide-gated cation channels regulate auditory coincidence detection in nucleus laminaris of the chick. J Neurosci, 2005; 25: 8867-8877.
- Kuba H, Yamada R, Fukui I, Ohmori H. Tonotopic specialization of auditory coincidence detection in nucleus laminaris of the chick. J Neurosci, 2005; 25: 1924-1934.
MESSAGE
We are recruiting graduate students (master and doctoral courses). For those who are interested in the above themes, please feel free to contact Dr. Kuba (kuba[AT]med.Nagoya-u.ac.jp)(please replace [AT] by @). Detailed information is available at the following site (https://www.med.nagoya-u.ac.jp/medical_E/graduate/).

