Molecular Biology
KEYWORDS
- Regulation of cancer cell proliferation
- Epigenetics
- Prolyl isomerases
- Calcium signaling
- Regulation of chromatin structure
HEAD

LAB MEMBER
| Faculty | Position | Researchers |
|---|---|---|
| TSUBOTA Shoma | Assistant Professor | Researchers |
| SAKAKIBARA Shotaro | Assistant Professor | Researchers |
| SUGIYAMA Shigeaki | Assistant Professor | Researchers |
CONTACT
| shimada◎med.nagoya-u.ac.jp(Please send a message after replacing "◎" mark with "@" mark. ) | |
| HP | Private Page |
OUTLINE
Our laboratory conducts integrative research ranging from elucidating the fundamental mechanisms of inheritance of genetic information and gene expression regulation—central to all biological phenomena—to the identification of therapeutic targets and the development of treatment strategies for cancer.
This page highlights our three major research themes:
- Identification of Therapeutic Targets and Development of Treatment Strategies in Refractory Cancers
- Understanding Biological Phenomena Mediated by Prolyl Isomerization
- Dysregulation of Calcium Signaling in Aberrant Cell Proliferation and Cancer
RESEARCH PROJECTS
1. Identification of Therapeutic Targets and Development of Treatment Strategies in Refractory Cancers
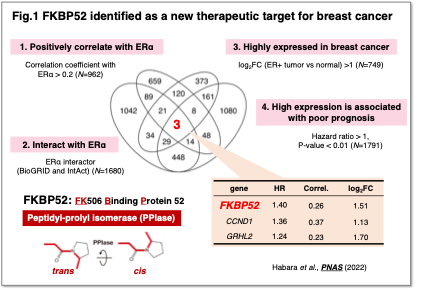
We aim to elucidate the molecular mechanisms underlying therapeutic resistance and cancer progression, with the goal of identifying novel therapeutic targets and developing new treatment strategies. As an example, we have explored potential therapeutic targets in estrogen receptor α (ERα)-positive breast cancer. Breast cancer is the most prevalent malignancy among Japanese women, and approximately 70% of cases are classified as ERα-positive. While endocrine therapy is effective for these tumors, resistance to treatment remains a major challenge. By analyzing molecules functionally associated with ERα and correlated with prognosis, we identified FKBP52 (FK506 Binding Protein 52) as a therapeutic target (Fig. 1)( Habara et al.,PNAS, 2022). Suppression of FKBP52 was found to promote ERα degradation and inhibit cancer cell proliferation, and notably, targeting FKBP52 was also effective in endocrine therapy–resistant breast cancer.
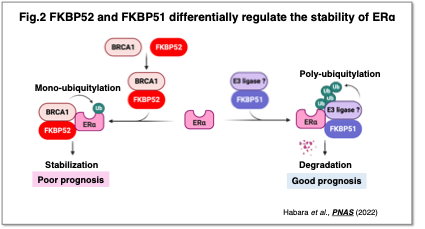
Moreover, we found that FKBP52 stabilizes ERα via interaction with BRCA1, whereas FKBP51, a homolog of FKBP52, facilitates ERα degradation and functions as a tumor suppressor (Fig. 2). Through comprehensive analysis of factors involved in cancer cell proliferation and therapeutic resistance, we are clarifying the molecular functions of candidate therapeutic targets and striving to translate these findings into personalized medicine and the development of innovative therapies.
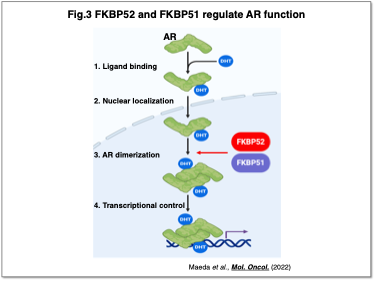
In prostate cancer, we have elucidated the roles of androgen receptor (AR) and the FKBP family, demonstrating that FKBP51 and FKBP52 are essential for (1) AR dimerization and (2) the expression of AR target genes (Fig. 3) (Maeda et al., Mol. Oncol., 2021). Inhibition of these FKBPs was effective even in castration-resistant prostate cancer cells, highlighting their potential as therapeutic targets. At present, we are investigating the molecular mechanisms underlying therapeutic resistance and malignancy in a variety of cancer types, with the aim of developing novel molecularly targeted therapies based on these insights.
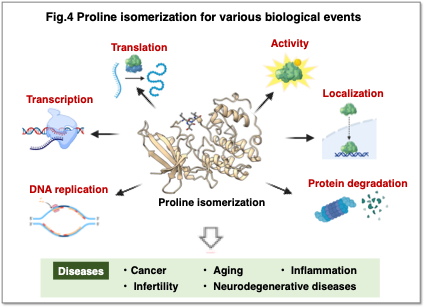
2. Understanding Biological Phenomena Mediated by Prolyl Isomerization
Prolyl isomerases have been implicated in various physiological processes, including DNA replication, transcription, translation, protein activity, localization, and degradation (Fig. 4). Dysregulation of prolyl isomerization is associated with a broad spectrum of diseases, such as cancer, aging, inflammation, infertility, and neurodegenerative disorders, and research in this field is rapidly advancing.
However, only a limited number of studies have addressed the extent to which prolyl isomerization affects protein conformation or the mechanisms by which it influences physiological functions. Although traditional studies in biology have focused on post-translational modifications such as phosphorylation and ubiquitination, recent evidence suggests that conformational changes induced by prolyl isomerization may also play a critical role in regulating complex biological processes. The physiological significance and molecular mechanisms underlying prolyl isomerization remain largely unexplored. We are currently conducting systematic analyses of the impact of prolyl isomerization on protein structure and the consequent effects on physiological function, employing both structural biology and cell biology approaches. Through this research, we seek to elucidate the biological significance of prolyl isomerization and to contribute to the understanding of disease mechanisms and the identification of novel therapeutic targets for conditions such as cancer and neurodegenerative diseases.
3. Dysregulation of Calcium Signaling in Aberrant Cell Proliferation and Cancer
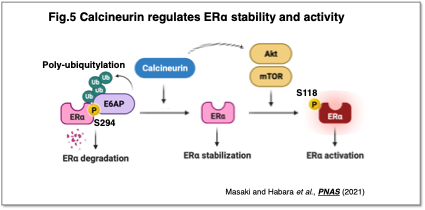
Calcineurin, a calcium-dependent protein phosphatase, is known as the molecular target of the immunosuppressant FK506. We have identified calcineurin as a novel regulatory factor in recurrent breast cancer (Masaki and Habara et al., PNAS, 2021) (Fig. 5). Calcineurin prevents the degradation of estrogen receptor α (ERα) through dephosphorylation and further promotes ERα activation via the mTOR pathway, thereby enhancing ERα function and contributing to cancer recurrence and poor prognosis.
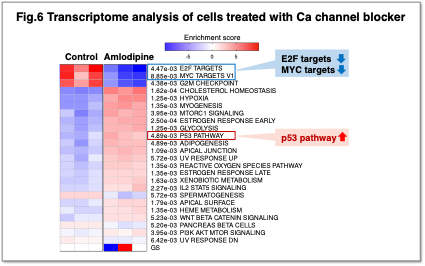
Intracellular calcium (Ca2+) is a critical second messenger involved in diverse cellular phenomena such as gene expression, cell cycle progression, and apoptosis. In cancer cells, disruption of Ca2+ homeostasis is thought to drive malignant transformation and metastasis. Through transcriptomic analysis using the Ca2+ channel blocker amlodipine, we found that reduced intracellular Ca2+ levels suppress the expression of c-Myc and E2F target genes, which promote cell cycle progression, and activate p53 target genes (Fig. 6).
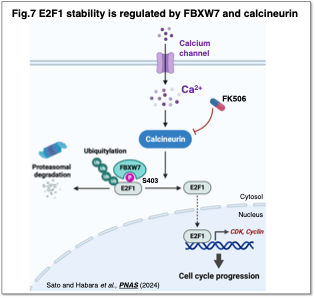
Regarding the regulation of E2F1 by intracellular Ca2+ concentration, we discovered that while the ubiquitin ligase FBXW7 promotes E2F1 degradation, calcineurin-mediated dephosphorylation inhibits this interaction and leads to E2F1 stabilization (Sato and Habara et al., PNAS, 2024) (Fig. 7).
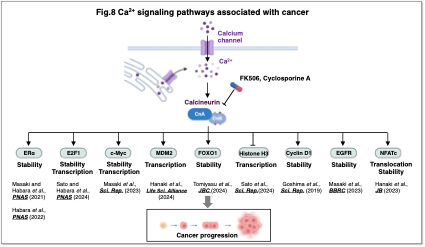
We further demonstrated that calcineurin stabilizes c-Myc and activates MDM2 transcription through nuclear translocation of NFATc (Masaki et al., Sci. Rep., 2023; Hanaki et al., Life Sci. Alliance, 2024). These findings underscore the potential of calcium signaling and calcineurin as therapeutic targets in cancer.
Aberrant calcium signaling is intimately associated with cancer initiation and progression, and we are actively investigating its molecular underpinnings. To date, we have identified several calcineurin substrates relevant to cancer progression (Fig. 8), and our ongoing research aims to develop innovative cancer therapies targeting calcium signaling pathways.
BIBLIOGRAPHY
2025
- Haba H, Tsubota S, Shimada M, Calcineurin in cancer signaling networks. Nagoya J Med Sci. 87, 182-195, 2025.
2024
- Sato Y, Habara M, Hanaki S, Masaki T, Tomiyasu H, Miki Y, Sakurai M, Morimoto M, Kobayashi D, Miyamoto T, Shimada M*: Calcineurin-mediated dephosphorylation stabilizes E2F1 protein by suppressing binding of the FBXW7 ubiquitin ligase subunit. PNAS, 2024 Oct 8;121(41):e2414618121.
- Sato Y, Habara M, Hanaki S, Sharif J, Tomiyasu H, Miki Y, Shimada M*: Calcineurin/NFATc1 pathway represses cellular cytotoxicity by modulating histone H3 expression. Sci Rep., 14, 14732, 2024.
- Hanaki S, Habara M, Tomiyasu H, Sato Y, Miki Y, Masaki T, Shibutani S, Shimada M*: NFAT activation by FKBP52 promotes cancer cell proliferation by suppressing p53. Life Sci Alliance., 7, e202302426, 2024.
- Tomiyasu H, Habara M, Hanaki S, Sato Y, Miki Y, Shimada M*: FOXO1 promotes cancer cell growth through MDM2-mediated p53 degradation. J. Biol. Chem.,300, 107209, 2024.
- Hanaki S, Habara M, Sato Y, Tomiyasu H, Miki Y, Shibutani S, Shimada M*: Dephosphorylation of NFAT by Calcineurin inhibits. Skp2-mediated degradation. J Biochem., 175, 235-244, 2024.
2023
- Masaki T, Habara M, Hanaki S, Sato Y, Tomiyasu H, Miki Y, Shimada M*: Calcineurin-mediated dephosphorylation enhances the stability and transactivation of c-Myc. Sci. Rep., 13, 13116, 2023.
- Masaki T, Habara M, Shibutani S, Hanaki S, Sato Y, Tomiyasu H, Shimada M*: Dephosphorylation of the EGFR protein by calcineurin at serine 1046/1047 enhances its stability. Biochem. Biophys. Res. Commun., 641, 84-92, 2023.
- Suzuki, Y., Kadomatsu, K., Sakamoto, K. Towards the in vivo identification of protein-protein interactions. J Biochem. mvad013 (2023).
- Jun Ouchida, Tomoya Ozaki, Naoki Segi, Yuji Suzuki, Shiro Imagama, Kenji Kadomatsu, Kazuma Sakamoto. Glypican-2 defines age-dependent axonal response to chondroitin sulfate. Exp Neurol. 2023 May 15.doi: 10.1016/j.expneurol.2023.114444.
2022
- Habara M, Sato Y, Goshima T, Sakurai M, Imai H, Shimizu H, Katayama Y, Hanaki S, Masaki T, Morimoto M, Nishikawa S, Toyama T, Shimada M*: FKBP52 and FKBP51 Differentially Regulate the Stability of Estrogen Receptor in Breast Cancer. PNAS, 119, e2110256119, 2022.
- Segi, N., Ozaki, T., Suzuki, Y., Ouchida, J., Imagama, S, Kadomatsu, K., Sakamoto, K. Close association of polarization and LC3, a marker of autophagy, in axon determination in mouse hippocampal neurons. Exp Neurol. 114112 (2022). doi: 10.1016/j.expneurol.2022.114112. Online ahead of print.
2021
- Masaki T, Habara M, Sato Y, Goshima T, Maeda K, Hanaki S, Shimada M*: Calcineurin regulates the stability and activity of estrogen receptor α. PNAS, 118, e2114258118, 2021.
- Maeda K, Habara M, Kawaguchi M, Matsumoto H, Hanaki S, Masaki T, Sato Y, Matsuyama H, Kunieda K, Nakagawa H, Shimada M*: FKBP51 and FKBP52 regulate androgen receptor dimerization and proliferation in prostate cancer cells. Mol Oncol., 16, 940-956, 2021. [Link]
- Hanaki S, Habara M, Masaki T, Maeda K, Sato Y, Nakanishi M, Shimada M*: PP1 regulatory subunit NIPP1 regulates transcription of E2F1 target genes following DNA damage. Cancer Sci., 112, 2739-2752, 2021.
- Machino M, Gong Y, Ozaki T, Suzuki Y, Watanabe E, Imagama S, Kadomatsu K, Sakamoto K. Dermatan sulfate is an activating ligand of anaplastic lymphoma kinase. J Biochem. 2021 Dec 28;170(5):631-637. doi: 10.1093/jb/mvab085. Online ahead of print. PMID: 34270745
- Gong Y, Abudureyimu S, Kadomatsu K, Sakamoto K. Identification of PTPRσ-interacting proteins by proximity-labelling assay. J Biochem. 2021 Mar 5;169(2):187-194. doi: 10.1093/jb/mvaa141.PMID: 33313879
- Ito S, Ozaki T, Morozumi M, Imagama S, Kadomatsu K, Sakamoto K. Enoxaparin promotes functional recovery after spinal cord injury by antagonizing PTPRσ. Exp. Neurol. 2021 Jun;340:113679. doi: 10.1016/j.expneurol.2021.113679. Epub 2021 Mar 1.
- Sakamoto K, Ozaki T, Suzuki Y, Kadomatsu K. Type IIa RPTPs and Glycans: Roles in Axon Regeneration and Synaptogenesis. Int J Mol Sci. 2021 May 24;22(11):5524. doi:10.3390/ijms22115524.PMID: 34073798
- Sakamoto K, Ozaki T, Kadomatsu K. Axonal Regeneration by Glycosaminoglycan. Front Cell Dev. Biol., 2021 Jun 16;9:702179. doi: 10.3389/fcell.2021.702179. eCollection 2021.
- Komata Y, Tsubota S, Sakamoto K, Ikematsu S, Kadomatsu K. Screening of novel Midkine binding protein by BioID2-based proximity labeling. Nagoya J Med
2020
- Mikawa T, Shibata E, Shimada M, Ito K, Ito T, Kanda H, Takubo K, Lleonart ME, Inagaki N, Yokode M, Kondoh H*: Phosphoglycerate mutase cooperates with Chk1 kinase to regulate glycolysis. iScience, 23, 101306, 2020.
- Yamashita K, Kiyonari S, Tsubota S, Kishida S, Sakai R, Kadomatsu K. Thymidylate synthase inhibitor raltitrexed can induce high levels of DNA damage in MYCN-amplified neuroblastoma cells. Cancer Sci. 2020
2019
- Nariai Y, Kamino H, Obayashi E, Kato H, Sakashita G, Sugiura T, Migita K, Koga T, Kawakami A, Sakamoto K, Kadomatsu K, Nakakido M, Tsumoto K, Urano T. Generation and characterization of antagonistic anti-human interleukin (IL)-18 monoclonal antibodies with high affinity: Two types of monoclonal antibodies against full-length IL-18 and the neoepitope of inflammatory caspase-cleaved active IL-18. Arch. Biochem. Biophys. 2019 Jan 4;663:71-82.
- Takeda-Okuda N, Mizumoto S, Zhang Z, Kim SK, Lee CH, Jeon BT, Hosaka YZ, Kadomatsu K, Yamada S, Tamura JI. Compositional analysis of the glycosaminoglycan family in velvet antlers of Sika deer (Cervus nippon) at different growing stages. Glycoconj. J. 2019 Jan 24.
- Funahashi Y, Kato N, Masuda T, Nishio F, Kitai H, Ishimoto T, Kosugi T, Tsuboi N, Matsuda N, Maruyama S, Kadomatsu K. miR-146a targeted to splenic macrophages prevents sepsis-induced multiple organ injury. Lab. Invest. 2019 Jan 30.
- Narentuya, Takeda-Uchimura Y, Foyez T, Zhang Z, Akama TO, Yagi H, Kato K, Komatsu Y, Kadomatsu K, Uchimura K. GlcNAc6ST3 is a keratan sulfate sulfotransferase for the protein-tyrosine phosphatase PTPRZ in the adult brain. Sci Rep. 2019 Mar 13;9(1):4387.
- Sakamoto K, Ozaki,T, Yen-Chun Ko, Cheng-Fang Tsai, Gong Y, Morozumi,M, Ishikawa, Y, Uchimura K, Nadanaka S, Kitagawa H, Medel Manuel L. Zulueta, Anandaraju Bandaru, Tamura J, Shang-Cheng Hung, Kadomatsu K. Glycan sulfation patterns define autophagy flux at axon tip via PTPRσ-cortactin axis. Nature Chemical Biology, 2019.
2018
- Doke T, Ishimoto T, Hayasaki T, Ikeda S, Hasebe M, Hirayama A, Soga T, Kato N, Kosugi T, Tsuboi N, Lanaspa MA, Johnson RJ, Kadomatsu K, Maruyama S. Lacking Ketohexokinase-A Exacerbates Renal Injury in Streptozotocin-induced Diabetic Mice. Metabolism, 2018.
- Tsubota S, Kadomatsu K. Origin and initiation mechanisms of neuroblastoma. Cell Tissue Res, 2018; 372: 211-221.
- Mori Y, Masuda T, Kosugi T, Yoshioka T, Hori M, Nagaya H, Maeda K, Sato Y, Kojima H, Kato N, Ishimoto T, Katsuno T, Yuzawa Y, Kadomatsu K, Maruyama S. The clinical relevance of plasma CD147/basigin in biopsy-proven kidney diseases. Clin Exp Nephrol, 2018; 22: 815-824.
2017
- Iwata T, Uchino T, Koyama A, Johmura Y, Koyama J, Saito T, Ishiguro S, Arikawa T, Komatsu S, Miyachi M, Sano T, Nakanishi M, Shimada M*: The G2 checkpoint inhibitor CBP-93872 increases the sensitivity of colorectal and pancreatic cancer cells to chemotherapy. PLoS ONE, 12, e0178221, 2017
- Su Z, Kishida S, Tsubota S, Sakamoto K, Cao D, Kiyonari S, Ohira M, Kamijo T, Narita A, Xu Y, Takahashi Y, Kadomatsu K. Neurocan, an extracellular chondroitin sulfate proteoglycan, stimulates neuroblastoma cells to promote malignant phenotypes. Oncotarget, 2017; 8: 106296-106310.
- Tsubota S, Kadomatsu K. Origin and mechanism of neuroblastoma. Oncoscience, 2017; 4: 70-72.
- Tsubota S, Kishida S, Shimamura T, Ohira M, Yamashita S, Cao D, Kiyonari S, Ushijima T, Kadomatsu K. PRC2-Mediated Transcriptomic Alterations at the Embryonic Stage Govern Tumorigenesis and Clinical Outcome in MYCN-Driven Neuroblastoma. Cancer Res, 2017; 77: 5259-5271.
- Takemoto Y, Horiba M, Harada M, Sakamoto K, Takeshita K, Murohara T, Kadomatsu K, Kamiya K. Midkine Promotes Atherosclerotic Plaque Formation Through Its Pro-Inflammatory, Angiogenic and Anti-Apoptotic Functions in Apolipoprotein E-Knockout Mice. Circ J, 2017; 82: 19-27.
- Tsubota S, Kadomatsu K. Neuroblastoma stem cells and CFC1. Oncotarget, 2017; 8: 45032-45033.
- Sakamoto K, Kadomatsu K. Mechanisms of axon regeneration: The significance of proteoglycans. Biochim Biophys Acta, 2017; 1861: 2435-2441.
- Ohgomori T, Yamasaki R, Takeuchi H, Kadomatsu K, Kira JI, Jinno S. Differential involvement of vesicular and glial glutamate transporters around spinal alpha-motoneurons in the pathogenesis of SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Neuroscience, 2017; 356: 114-124.
- Scilabra SD, Yamamoto K, Pigoni M, Sakamoto K, Muller SA, Papadopoulou A, Lichtenthaler SF, Troeberg L, Nagase H, Kadomatsu K. Dissecting the interaction between tissue inhibitor of metalloproteinases-3 (TIMP-3) and low density lipoprotein receptor-related protein-1 (LRP-1): Development of a \"TRAP\" to increase levels of TIMP-3 in the tissue. Matrix Biol, 2017; 59: 69-79.
2016
- Ho WL, Hsu WM, Huang MC, Kadomatsu K, Nakagawara A. Protein glycosylation in cancers and its potential therapeutic applications in neuroblastoma. J Hematol Oncol, 2016; 9: 100.
- Ukai J, Imagama S, Ohgomori T, Ito Z, Ando K, Ishiguro N, Kadomatsu K. Nogo receptor 1 is expressed in both primary cultured glial cells and neurons. Nagoya J Med Sci, 2016; 78: 303-311.
- Zhang Z, Ohtake-Niimi S, Kadomatsu K, Uchimura K. Reduced molecular size and altered disaccharide composition of cerebral chondroitin sulfate upon Alzheimer's pathogenesis in mice. Nagoya J Med Sci, 2016; 78: 293-301.
- Murakami-Tonami Y, Ikeda H, Yamagishi R, Inayoshi M, Inagaki S, Kishida S, Komata Y, Jan K, Takeuchi I, Kondo Y, Maeda T, Sekido Y, Murakami H, Kadomatsu K. SGO1 is involved in the DNA damage response in MYCN-amplified neuroblastoma cells. Sci Rep, 2016; 6: 31615.
- Hayashi H, Sato W, Kosugi T, Nishimura K, Sugiyama D, Asano N, Ikematsu S, Komori K, Nishiwaki K, Kadomatsu K, Matsuo S, Maruyama S, Yuzawa Y. Efficacy of urinary midkine as a biomarker in patients with acute kidney injury. Clin Exp Nephrol, 2016.
- Honda Y, Shishido T, Takahashi T, Watanabe T, Netsu S, Kinoshita D, Narumi T, Kadowaki S, Nishiyama S, Takahashi H, Arimoto T, Miyamoto T, Kishida S, Kadomatsu K, Takeishi Y, Kubota I. Midkine Deteriorates Cardiac Remodeling via Epidermal Growth Factor Receptor Signaling in Chronic Kidney Disease. Hypertension, 2016; 67: 857-865.
- Ohgomori T, Yamada J, Takeuchi H, Kadomatsu K, Jinno S. Comparative morphometric analysis of microglia in the spinal cord of SOD1(G93A) transgenic mouse model of amyotrophic lateral sclerosis. Eur J Neurosci, 2016; 43: 1340-1351.
- Suzuki K, Satoh K, Ikeda S, Sunamura S, Otsuki T, Satoh T, Kikuchi N, Omura J, Kurosawa R, Nogi M, Numano K, Sugimura K, Aoki T, Tatebe S, Miyata S, Mukherjee R, Spinale FG, Kadomatsu K, Shimokawa H. Basigin Promotes Cardiac Fibrosis and Failure in Response to Chronic Pressure Overload in Mice. Arterioscler Thromb Vasc Biol, 2016; 36: 636-646.
- Hashimoto H, Ishino Y, Jiang W, Yoshimura T, Takeda-Uchimura Y, Uchimura K, Kadomatsu K, Ikenaka K. Keratan Sulfate Regulates the Switch from Motor Neuron to Oligodendrocyte Generation During Development of the Mouse Spinal Cord. Neurochem Res, 2016; 41: 450-462.
2015
- Kamiguchi H, Kadomatsu K. Introduction to glyco-neuroscience. Exp Neurol, 2015; 274: 89.
- Fujimoto H, Ohgomori T, Abe K, Uchimura K, Kadomatsu K, Jinno S. Time-dependent localization of high- and low-sulfated keratan sulfates in the song nuclei of developing zebra finches. Eur J Neurosci, 2015; 42: 2716-2725.
- Foyez T, Takeda-Uchimura Y, Ishigaki S, Narentuya, Zhang Z, Sobue G, Kadomatsu K, Uchimura K. Microglial keratan sulfate epitope elicits in central nervous tissues of transgenic model mice and patients with amyotrophic lateral sclerosis. Am J Pathol, 2015; 185: 3053-3065.
- Ueno R, Miyamoto K, Tanaka N, Moriguchi K, Kadomatsu K, Kusunoki S. Keratan sulfate exacerbates experimental autoimmune encephalomyelitis. J Neurosci Res, 2015; 93: 1874-1880.
- Takeda-Uchimura Y, Uchimura K, Sugimura T, Yanagawa Y, Kawasaki T, Komatsu Y, Kadomatsu K. Requirement of keratan sulfate proteoglycan phosphacan with a specific sulfation pattern for critical period plasticity in the visual cortex. Exp Neurol, 2015; 274: 145-155.
- Kiyonari S, Iimori M, Matsuoka K, Watanabe S, Morikawa-Ichinose T, Miura D, Niimi S, Saeki H, Tokunaga E, Oki E, Morita M, Kadomatsu K, Maehara Y, Kitao H. The 1,2-Diaminocyclohexane Carrier Ligand in Oxaliplatin Induces p53-Dependent Transcriptional Repression of Factors Involved in Thymidylate Biosynthesis. Mol Cancer Ther, 2015; 14: 2332-2342.
- Maeda K, Kosugi T, Sato W, Kojima H, Sato Y, Kamimura D, Kato N, Tsuboi N, Yuzawa Y, Matsuo S, Murakami M, Maruyama S, Kadomatsu K. CD147/basigin limits lupus nephritis and Th17 cell differentiation in mice by inhibiting the interleukin-6/STAT-3 pathway. Arthritis Rheumatol, 2015; 67: 2185-2195.
- Matsuda Y, Haneda M, Kadomatsu K, Kobayashi T. A proliferation-inducing ligand sustains the proliferation of human naive (CD27(-)) B cells and mediates their differentiation into long-lived plasma cells in vitro via transmembrane activator and calcium modulator and cyclophilin ligand interactor and B-cell mature antigen. Cell Immunol, 2015; 295: 127-136.
- Arima H, Omura T, Hayasaka T, Masaki N, Hanada M, Xu D, Banno T, Kobayashi K, Takeuchi H, Kadomatsu K, Matsuyama Y, Setou M. Reductions of docosahexaenoic acid-containing phosphatidylcholine levels in the anterior horn of an ALS mouse model. Neuroscience, 2015; 297: 127-136.
- Chen D, Ito S, Yuan H, Hyodo T, Kadomatsu K, Hamaguchi M, Senga T. EML4 promotes the loading of NUDC to the spindle for mitotic progression. Cell Cycle, 2015; 14: 1529-1539.
- Ishikawa Y, Imagama S, Ohgomori T, Ishiguro N, Kadomatsu K. A combination of keratan sulfate digestion and rehabilitation promotes anatomical plasticity after rat spinal cord injury. Neurosci Lett, 2015; 593: 13-18.
- Yuan Y, Makita N, Cao D, Mihara K, Kadomatsu K, Takei Y. Atelocollagen-mediated intravenous siRNA delivery specific to tumor tissues orthotopically xenografted in prostates of nude mice and its anticancer effects. Nucleic Acid Ther, 2015; 25: 85-94.
- Lu F, Kishida S, Mu P, Huang P, Cao D, Tsubota S, Kadomatsu K. NeuroD1 promotes neuroblastoma cell growth by inducing the expression of ALK. Cancer Sci, 2015; 106: 390-396.
- Nakaguro M, Kiyonari S, Kishida S, Cao D, Murakami-Tonami Y, Ichikawa H, Takeuchi I, Nakamura S, Kadomatsu K. Nucleolar protein PES1 is a marker of neuroblastoma outcome and is associated with neuroblastoma differentiation. Cancer Sci, 2015; 106: 237-243.
- Sato Y, Sato W, Maruyama S, Wilcox CS, Falck JR, Masuda T, Kosugi T, Kojima H, Maeda K, Furuhashi K, Ando M, Imai E, Matsuo S, Kadomatsu K. Midkine Regulates BP through Cytochrome P450-Derived Eicosanoids. J Am Soc Nephrol, 2015; 26: 1806-1815.
- Kiyonari S, Kadomatsu K. Neuroblastoma models for insights into tumorigenesis and new therapies. Expert Opin Drug Discov, 2015; 10: 53-62.
- Kosugi T, Maeda K, Sato W, Maruyama S, Kadomatsu K. CD147 (EMMPRIN/Basigin) in kidney diseases: from an inflammation and immune system viewpoint. Nephrol Dial Transplant, 2015; 30: 1097-1103.
2014
- Satoh K, Satoh T, Kikuchi N, Omura J, Kurosawa R, Suzuki K, Sugimura K, Aoki T, Nochioka K, Tatebe S, Miyamichi-Yamamoto S, Miura M, Shimizu T, Ikeda S, Yaoita N, Fukumoto Y, Minami T, Miyata S, Nakamura K, Ito H, Kadomatsu K, Shimokawa H. Basigin mediates pulmonary hypertension by promoting inflammation and vascular smooth muscle cell proliferation. Circ Res, 2014; 115: 738-750.
- Shinjo R, Imagama S, Ito Z, Ando K, Nishida Y, Ishiguro N, Kadomatsu K. Keratan sulfate expression is associated with activation of a subpopulation of microglia/macrophages in Wallerian degeneration. Neurosci Lett, 2014; 579: 80-85.
- Moreno V, Gonzalo P, Gomez-Escudero J, Pollan A, Acin-Perez R, Breckenridge M, Yanez-Mo M, Barreiro O, Orsenigo F, Kadomatsu K, Chen CS, Enriquez JA, Dejana E, Sanchez-Madrid F, Arroyo AG. An EMMPRIN-gamma-catenin-Nm23 complex drives ATP production and actomyosin contractility at endothelial junctions. J Cell Sci, 2014; 127: 3768-3781.
- Kadomatsu K, Sakamoto K. Mechanisms of axon regeneration and its inhibition: roles of sulfated glycans. Arch Biochem Biophys, 2014; 558: 36-41.
- Miyamoto K, Tanaka N, Moriguchi K, Ueno R, Kadomatsu K, Kitagawa H, Kusunoki S. Chondroitin 6-O-sulfate ameliorates experimental autoimmune encephalomyelitis. Glycobiology, 2014; 24: 469-475.
- Murakami-Tonami Y, Kishida S, Takeuchi I, Katou Y, Maris JM, Ichikawa H, Kondo Y, Sekido Y, Shirahige K, Murakami H, Kadomatsu K. Inactivation of SMC2 shows a synergistic lethal response in MYCN-amplified neuroblastoma cells. Cell Cycle, 2014; 13: 1115-1131.
- Maeda-Hori M, Kosugi T, Kojima H, Sato W, Inaba S, Maeda K, Nagaya H, Sato Y, Ishimoto T, Ozaki T, Tsuboi N, Muro Y, Yuzawa Y, Imai E, Johnson RJ, Matsuo S, Kadomatsu K, Maruyama S. Plasma CD147 reflects histological features in patients with lupus nephritis. Lupus, 2014; 23: 342-352.
- Cao D, Kishida S, Huang P, Mu P, Tsubota S, Mizuno M, Kadomatsu K. A new tumorsphere culture condition restores potentials of self-renewal and metastasis of primary neuroblastoma in a mouse neuroblastoma model. PLoS One, 2014; 9: e86813.
- Muramatsu T, Kadomatsu K. Midkine: an emerging target of drug development for treatment of multiple diseases. Br J Pharmacol, 2014; 171: 811-813.
- Nagaya H, Kosugi T, Maeda-Hori M, Maeda K, Sato Y, Kojima H, Hayashi H, Kato N, Ishimoto T, Sato W, Yuzawa Y, Matsuo S, Kadomatsu K, Maruyama S. CD147/basigin reflects renal dysfunction in patients with acute kidney injury. Clin Exp Nephrol, 2014; 18: 746-754.
- Kadomatsu K, Bencsik P, Gorbe A, Csonka C, Sakamoto K, Kishida S, Ferdinandy P. Therapeutic potential of midkine in cardiovascular disease. Br J Pharmacol, 2014; 171: 936-944.
- Kadomatsu K, Sakamoto K. Sulfated glycans in network rewiring and plasticity after neuronal injuries. Neurosci Res, 2014; 78: 50-54.
- Hoshino H, Foyez T, Ohtake-Niimi S, Takeda-Uchimura Y, Michikawa M, Kadomatsu K, Uchimura K. KSGal6ST is essential for the 6-sulfation of galactose within keratan sulfate in early postnatal brain. J Histochem Cytochem, 2014; 62: 145-156.
- Kishida S, Kadomatsu K. Involvement of midkine in neuroblastoma tumourigenesis. Br J Pharmacol, 2014; 171: 896-904.
2013
- Hasan MK, Nafady A, Takatori A, Kishida S, Ohira M, Suenaga Y, Hossain S, Akter J, Ogura A, Nakamura Y, Kadomatsu K, Nakagawara A. ALK is a MYCN target gene and regulates cell migration and invasion in neuroblastoma. Sci Rep, 2013; 3: 3450.
- Matsui H, Ohgomori T, Natori T, Miyamoto K, Kusunoki S, Sakamoto K, Ishiguro N, Imagama S, Kadomatsu K. Keratan sulfate expression in microglia is diminished in the spinal cord in experimental autoimmune neuritis. Cell Death Dis, 2013; 4: e946.
- Duverle DA, Takeuchi I, Murakami-Tonami Y, Kadomatsu K, Tsuda K. Discovering combinatorial interactions in survival data. Bioinformatics, 2013; 29: 3053-3059.
- Hirano K, Ohgomori T, Kobayashi K, Tanaka F, Matsumoto T, Natori T, Matsuyama Y, Uchimura K, Sakamoto K, Takeuchi H, Hirakawa A, Suzumura A, Sobue G, Ishiguro N, Imagama S, Kadomatsu K. Ablation of keratan sulfate accelerates early phase pathogenesis of ALS. PLoS One, 2013; 8: e66969.
- Muramoto A, Imagama S, Natori T, Wakao N, Ando K, Tauchi R, Hirano K, Shinjo R, Matsumoto T, Ishiguro N, Kadomatsu K. Midkine overcomes neurite outgrowth inhibition of chondroitin sulfate proteoglycan without glial activation and promotes functional recovery after spinal cord injury. Neurosci Lett, 2013; 550: 150-155.
- Kadomatsu K, Kishida S, Tsubota S. The heparin-binding growth factor midkine: the biological activities and candidate receptors. J Biochem, 2013; 153: 511-521.
- Kobayashi K, Imagama S, Ohgomori T, Hirano K, Uchimura K, Sakamoto K, Hirakawa A, Takeuchi H, Suzumura A, Ishiguro N, Kadomatsu K. Minocycline selectively inhibits M1 polarization of microglia. Cell Death Dis, 2013; 4: e525.
- Kishida S, Mu P, Miyakawa S, Fujiwara M, Abe T, Sakamoto K, Onishi A, Nakamura Y, Kadomatsu K. Midkine promotes neuroblastoma through Notch2 signaling. Cancer Res, 2013; 73: 1318-1327.
- Kojima H, Kosugi T, Sato W, Sato Y, Maeda K, Kato N, Kato K, Inaba S, Ishimoto T, Tsuboi N, Matsuo S, Maruyama S, Yuzawa Y, Kadomatsu K. Deficiency of growth factor midkine exacerbates necrotizing glomerular injuries in progressive glomerulonephritis. Am J Pathol, 2013; 182: 410-419.
2012
- Sakamoto K, Kadomatsu K. Midkine in the pathology of cancer, neural disease, and inflammation. Pathol Int, 2012; 62: 445-455.
- Matsumoto T, Imagama S, Hirano K, Ohgomori T, Natori T, Kobayashi K, Muramoto A, Ishiguro N, Kadomatsu K. CD44 expression in astrocytes and microglia is associated with ALS progression in a mouse model. Neurosci Lett, 2012; 520: 115-120.
- Koide N, Yasuda K, Kadomatsu K, Takei Y. Establishment and optimal culture conditions of microrna-induced pluripotent stem cells generated from HEK293 cells via transfection of microrna-302s expression vector. Nagoya J Med Sci, 2012; 74: 157-165.
- Tauchi R, Imagama S, Ohgomori T, Natori T, Shinjo R, Ishiguro N, Kadomatsu K. ADAMTS-13 is produced by glial cells and upregulated after spinal cord injury. Neurosci Lett, 2012; 517: 1-6.
- Tauchi R, Imagama S, Natori T, Ohgomori T, Muramoto A, Shinjo R, Matsuyama Y, Ishiguro N, Kadomatsu K. The endogenous proteoglycan-degrading enzyme ADAMTS-4 promotes functional recovery after spinal cord injury. J Neuroinflammation, 2012; 9: 53.
- Sonobe Y, Li H, Jin S, Kishida S, Kadomatsu K, Takeuchi H, Mizuno T, Suzumura A. Midkine inhibits inducible regulatory T cell differentiation by suppressing the development of tolerogenic dendritic cells. J Immunol, 2012; 188: 2602-2611.
- Sakai K, Yamamoto A, Matsubara K, Nakamura S, Naruse M, Yamagata M, Sakamoto K, Tauchi R, Wakao N, Imagama S, Hibi H, Kadomatsu K, Ishiguro N, Ueda M. Human dental pulp-derived stem cells promote locomotor recovery after complete transection of the rat spinal cord by multiple neuro-regenerative mechanisms. J Clin Invest, 2012; 122: 80-90.
- Inaba S, Nagahara S, Makita N, Tarumi Y, Ishimoto T, Matsuo S, Kadomatsu K, Takei Y. Atelocollagen-mediated systemic delivery prevents immunostimulatory adverse effects of siRNA in mammals. Mol Ther, 2012; 20: 356-366.
2011
- Imagama S, Sakamoto K, Tauchi R, Shinjo R, Ohgomori T, Ito Z, Zhang H, Nishida Y, Asami N, Takeshita S, Sugiura N, Watanabe H, Yamashita T, Ishiguro N, Matsuyama Y, Kadomatsu K. Keratan sulfate restricts neural plasticity after spinal cord injury. J Neurosci, 2011; 31: 17091-17102.
- Huet E, Vallee B, Delbe J, Mourah S, Pruliere-Escabasse V, Tremouilleres M, Kadomatsu K, Doan S, Baudouin C, Menashi S, Gabison EE. EMMPRIN modulates epithelial barrier function through a MMP-mediated occludin cleavage: implications in dry eye disease. Am J Pathol, 2011; 179: 1278-1286.
- Ishiguro H, Horiba M, Takenaka H, Sumida A, Opthof T, Ishiguro YS, Kadomatsu K, Murohara T, Kodama I. A single intracoronary injection of midkine reduces ischemia/reperfusion injury in Swine hearts: a novel therapeutic approach for acute coronary syndrome. Front Physiol, 2011; 2: 27.
- Hayashi M, Kadomatsu K, Kojima T, Ishiguro N. Keratan sulfate and related murine glycosylation can suppress murine cartilage damage in vitro and in vivo. Biochem Biophys Res Commun, 2011; 409: 732-737.
- Kadomatsu K. [Proteoglycans and neural circuit reconstruction]. Seikagaku, 2011; 83: 240-246.
- Kato K, Kosugi T, Sato W, Arata-Kawai H, Ozaki T, Tsuboi N, Ito I, Tawada H, Yuzawa Y, Matsuo S, Kadomatsu K, Maruyama S. Growth factor Midkine is involved in the pathogenesis of renal injury induced by protein overload containing endotoxin. Clin Exp Nephrol, 2011; 15: 346-354.
- Huang P, Kishida S, Cao D, Murakami-Tonami Y, Mu P, Nakaguro M, Koide N, Takeuchi I, Onishi A, Kadomatsu K. The neuronal differentiation factor NeuroD1 downregulates the neuronal repellent factor Slit2 expression and promotes cell motility and tumor formation of neuroblastoma. Cancer Res, 2011; 71: 2938-2948.
- Kato N, Kosugi T, Sato W, Ishimoto T, Kojima H, Sato Y, Sakamoto K, Maruyama S, Yuzawa Y, Matsuo S, Kadomatsu K. Basigin/CD147 promotes renal fibrosis after unilateral ureteral obstruction. Am J Pathol, 2011; 178: 572-579.
- Sakamoto K, Bu G, Chen S, Takei Y, Hibi K, Kodera Y, McCormick LM, Nakao A, Noda M, Muramatsu T, Kadomatsu K. Premature ligand-receptor interaction during biosynthesis limits the production of growth factor midkine and its receptor LDL receptor-related protein 1. J Biol Chem, 2011; 286: 8405-8413.
- Wakao N, Imagama S, Zhang H, Tauchi R, Muramoto A, Natori T, Takeshita S, Ishiguro N, Matsuyama Y, Kadomatsu K. Hyaluronan oligosaccharides promote functional recovery after spinal cord injury in rats. Neurosci Lett, 2011; 488: 299-304.
2010
- Hayashi M, Kadomatsu K, Ishiguro N. Keratan sulfate suppresses cartilage damage and ameliorates inflammation in an experimental mice arthritis model. Biochem Biophys Res Commun, 2010; 401: 463-468.
- Ito Z, Sakamoto K, Imagama S, Matsuyama Y, Zhang H, Hirano K, Ando K, Yamashita T, Ishiguro N, Kadomatsu K. N-acetylglucosamine 6-O-sulfotransferase-1-deficient mice show better functional recovery after spinal cord injury. J Neurosci, 2010; 30: 5937-5947.
- Kadomatsu K. Midkine regulation of the renin-angiotensin system. Curr Hypertens Rep, 2010; 12: 74-79.
- Asano Y, Kishida S, Mu P, Sakamoto K, Murohara T, Kadomatsu K. DRR1 is expressed in the developing nervous system and downregulated during neuroblastoma carcinogenesis. Biochem Biophys Res Commun, 2010; 394: 829-835.
- Miwa Y, Yamamoto K, Onishi A, Iwamoto M, Yazaki S, Haneda M, Iwasaki K, Liu D, Ogawa H, Nagasaka T, Uchida K, Nakao A, Kadomatsu K, Kobayashi T. Potential value of human thrombomodulin and DAF expression for coagulation control in pig-to-human xenotransplantation. Xenotransplantation, 2010; 17: 26-37.
- Sumida A, Horiba M, Ishiguro H, Takenaka H, Ueda N, Ooboshi H, Opthof T, Kadomatsu K, Kodama I. Midkine gene transfer after myocardial infarction in rats prevents remodelling and ameliorates cardiac dysfunction. Cardiovasc Res, 2010; 86: 113-121.
2009
- Hobo A, Yuzawa Y, Kosugi T, Kato N, Asai N, Sato W, Maruyama S, Ito Y, Kobori H, Ikematsu S, Nishiyama A, Matsuo S, Kadomatsu K. The growth factor midkine regulates the renin-angiotensin system in mice. J Clin Invest, 2009; 119: 1616-1625.
- Kato N, Yuzawa Y, Kosugi T, Hobo A, Sato W, Miwa Y, Sakamoto K, Matsuo S, Kadomatsu K. The E-selectin ligand basigin/CD147 is responsible for neutrophil recruitment in renal ischemia/reperfusion. J Am Soc Nephrol, 2009; 20: 1565-1576.
- Sakakima H, Yoshida Y, Yamazaki Y, Matsuda F, Ikutomo M, Ijiri K, Muramatsu H, Muramatsu T, Kadomatsu K. Disruption of the midkine gene (Mdk) delays degeneration and regeneration in injured peripheral nerve. J Neurosci Res, 2009; 87: 2908-2915.
- Mu P, Nagahara S, Makita N, Tarumi Y, Kadomatsu K, Takei Y. Systemic delivery of siRNA specific to tumor mediated by atelocollagen: combined therapy using siRNA targeting Bcl-xL and cisplatin against prostate cancer. Int J Cancer, 2009; 125: 2978-2990.
- Yin J, Sakamoto K, Zhang H, Ito Z, Imagama S, Kishida S, Natori T, Sawada M, Matsuyama Y, Kadomatsu K. Transforming growth factor-beta1 upregulates keratan sulfate and chondroitin sulfate biosynthesis in microglias after brain injury. Brain Res, 2009; 1263: 10-22.
- Takenaka H, Horiba M, Ishiguro H, Sumida A, Hojo M, Usui A, Akita T, Sakuma S, Ueda Y, Kodama I, Kadomatsu K. Midkine prevents ventricular remodeling and improves long-term survival after myocardial infarction. Am J Physiol Heart Circ Physiol, 2009; 296: H462-469.
- Kobayashi T, Liu D, Ogawa H, Miwa Y, Nagasaka T, Maruyama S, Li YT, Onishi A, Iwamoto M, Kuzuya T, Kadomatsu K, Uchida K, Nakao A. Removal of blood group A/B antigen in organs by ex vivo and in vivo administration of endo-beta-galactosidase (ABase) for ABO-incompatible transplantation. Transpl Immunol, 2009; 20: 132-138.
We welcome the master course and doctor course students

