Obstetrics and Gynecology
KEYWORDS
- Exosome
- pregnancy complication
- endometriosis
- Choriocarcinoma
- peritoneal mesothelium
- plasma
- placenta accreta spectrum
- RPOC
- PCOS
- reproductive axis
HEAD

LAB MEMBER
| Faculty | Position | Researchers |
|---|---|---|
| NAKAMURA Tomoko | Clinical Professor | Researchers |
| USHIDA Takafumi | Associate Professor | Researchers |
| NIIMI Kaoru | Associate Professor | Researchers |
| YOSHIKAWA Nobuhisa | Lecturer | Researchers |
| YOKOI Akira | Lecturer | Researchers |
| MURAOKA Ayako | Lecturer | Researchers |
| TAMAUCHI Satoshi | Clinical Lecturer | Researchers |
| YOSHIHARA Masato | Clinical Lecturer | Researchers |
| MIYAKE Natsuki | Assistant Professor | Researchers |
| TANO Sho | Assistant Professor | Researchers |
| SONEHARA Reina | Assistant Professor | Researchers |
| NAGAO Yukari | Assistant Professor | Researchers |
| KATSUKI Satoru | Assistant Professor | Researchers |
| YOSHIDA Kosuke | Assistant Clinical Professor | Researchers |
| MATSUO Seiko | Assistant Clinical Professor | Researchers |
| FUMA Kazuya | Assistant Clinical Professor | Researchers |
| SEKI Tomomi | Designated Assistant Professor | Researchers |
| MOGI Kazumasa | Assistant Clinical Professor | Researchers |
| IYOSHI Shohei | Designated Assistant Professor | Researchers |
CONTACT
| ob-gy◎med.nagoya-u.ac.jp (Please send a message after replacing "◎" mark with "@" mark. ) | |
| HP | Private Page |
OUTLINE
The Department of Obstetrics and Gynecology at Nagoya University conducts research through four specialized groups: Gynecologic Oncology, Endocrinology and Oncology, Perinatology, and Infertility and Reproductive Medicine.
RESEARCH PROJECTS
Research Interests – Oncology
Elucidation of the Mechanisms of Peritoneal Dissemination in Ovarian Cancer Focusing on Cancer-Associated Peritoneal Mesothelial Cells and the Development of Novel Therapeutic Strategies
Peritoneal mesothelial cells are recognized as key host cells involved in peritoneal dissemination of ovarian cancer. Under physiological conditions, these cells exhibit a cobblestone-like morphology and cover the surface of the visceral and parietal peritoneum, thereby reducing friction with intra-abdominal organs, producing and secreting various bioactive substances, and contributing to wound healing within the peritoneal cavity. This raises an important question: during ovarian cancer dissemination, do peritoneal mesothelial cells function in a “defensive” manner, or do they instead facilitate cancer progression by serving as a scaffold for the “initial contact” of cancer cells?
Recent studies have demonstrated that various cell types composing the tumor microenvironment secrete diverse growth factors and bioactive molecules that regulate therapeutic resistance, tumor growth, invasion, and metastasis. Among these cell populations, cancer-associated fibroblasts (CAFs) have been identified as a major contributor. We have previously demonstrated that factors derived from ovarian cancer cells induce phenotypic alterations in normally protective peritoneal mesothelial cells, transforming them into ovarian cancer-associated mesothelial cells (OCAMs) characterized by high α-SMA expression. The essence of OCAMs lies in epithelial–mesenchymal transition (EMT) of mesothelial cells.
We further revealed that OCAMs disrupt the blood–peritoneal barrier—composed of capillary lymphatic and blood vessel networks extending approximately 90 μm beneath the basal membrane—markedly increasing vascular permeability and contributing to massive ascites formation. Targeting OCAMs was shown to reduce ascites volume and improve the pathological condition of malignant peritonitis (Fujikake K et al., Oncology Reports, 2018).
In addition to mesothelial cells, immune cells and adipocytes are now recognized as intraperitoneal factors that promote ovarian cancer progression. Together, these components form a complex peritoneal ecosystem that plays a critical role in malignant peritonitis. A key event in this process is the acquisition of mesenchymal traits by tumor cells and the initiation and amplification of coordinated cancer–peritoneum cellular crosstalk. Indeed, mesothelial cells that transformed to OCAMs exhibit enhanced secretion of specific chemokines, actively recruiting ovarian cancer cells that highly express the corresponding chemokine receptors toward the peritoneum. This “seed and soil” mechanism increases peritoneal dissemination (Peng Y et al., Cancer Letters, 2019; Yasui H et al., Clinical & Experimental Metastasis, 2020). Furthermore, OCAMs cooperate with mesenchymal-type ovarian cancer cells to promote intraperitoneal platinum resistance (Yoshihara M et al., International Journal of Cancer, 2020).
Based on these findings, we believe that effective control of peritoneal dissemination requires strategies targeting the entire peritoneum to reprogram OCAMs back into normal mesothelial cells. In line with this concept, we have already identified several candidate compounds capable of turning OCAMs back to normal mesothelial cells and are pursuing novel therapeutic strategies aimed at controlling malignant peritonitis through “normalization of the peritoneal environment.”
Functional Analysis and Clinical Application of Cancer-Derived Exosomes in Gynecologic Malignancies
Exosomes are small lipid bilayer vesicles approximately 100 nm in diameter that are secreted by virtually all living cells. Since around 2007, they have been in the spotlight as a novel mechanism of intercellular communication. Exosomes are a subtype of extracellular vesicles (EVs), which are complex structures encapsulating various bioactive molecules. EVs play crucial roles in carcinogenesis, metastasis, and malignant progression, circulate abundantly in human body fluids, and dynamically change their molecular profiles in accordance with pathological conditions.
EV research is currently one of the most rapidly expanding fields worldwide, accompanied by remarkable growth in the surrounding market. Our research group has focused primarily on ovarian cancer, a gynecologic malignancy with poor prognosis, and has conducted extensive collaborative research with the National Cancer Center of Japan to elucidate the functional significance of exosomes. In 2017, we were the first to demonstrate that exosomes secreted by ovarian cancer cells contain MMP1 mRNA, which disrupts peritoneal mesothelial cells and promotes peritoneal dissemination (Yokoi A et al., Nature Communications, 2017).
Subsequently, as a part of the NEDO project “Development of Diagnostic Technology for Detection of miRNA in Body Fluids” we analyzed ten exosomal microRNAs in the blood of ovarian cancer patients and established an algorithm for early diagnosis (Yokoi A et al., Nature Communications, 2018). In collaboration with The University of Texas MD Anderson Cancer Center, we also elucidated the biosynthetic mechanisms of DNA contained within ovarian cancer-derived exosomes (Yokoi A et al., Science Advances, 2019).
Through comprehensive analyses of various nucleic acids contained in EVs and exosomes, we have confirmed their strong involvement in cancer malignant progression, as well as their potential as clinical biomarkers and novel therapeutic tools. We continue to conduct collaborative research with leading medical and research institutions nationwide. In addition, we are pursuing disease mechanism studies based on next-generation sequencing-driven comprehensive microRNA profiling and developing unique translational research projects targeting diseases encountered specifically in obstetrics and gynecology (Yoshida et al., Carcinogenesis, 2019; Yoshida et al., Oncogene, 2021).
With support from the Ministry of Education, Culture, Sports, Science and Technology’s Program for Forming Japan's Peak Research Universities, we established the “Nanotechnology based advanced approaches for cancer extracellular vesicles” at the Nagoya University Institute for Advanced Research. Through interdisciplinary collaboration with engineering fields and support from AMED programs, we are conducting cutting-edge research aimed at clinical translation that could result in social contribution of EV-based technologies.
Basic Research Aimed at the Application of Non-Equilibrium Atmospheric Pressure Plasma for Ovarian Cancer Treatment
In recent years, research on “plasma medicine,” which applies atmospheric pressure plasma to the human body for the treatment of diseases and injuries, has been actively pursued worldwide. While atmospheric pressure plasma has traditionally used for to environmental gas pollution purification/processing/management water purification, and sterilization, recent studies have extended its application to hemostasis, wound healing, and cancer therapy.
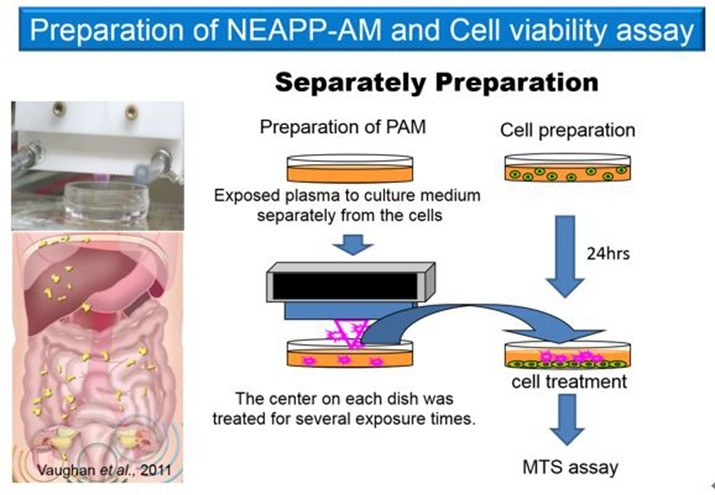
The therapeutic principle of plasma medicine involves reactive species, that directly attack cancer cells or moderately stimulate injured tissues to accelerate healing. At the cellular level, numerous studies have reported significant effects, and several animal studies are currently underway. Over the past decade, plasma irradiation has demonstrated to induce apoptosis not only in normal cells but also in cancer cells, including brain tumors, colorectal cancer, skin cancer, lung cancer, pancreatic cancer, and ovarian cancer.
This plasma-induced apoptosis is hypothesized to result from DNA damage caused by disruption of cellular redox balance due to reactive oxygen species (ROS), including free radicals generated by plasma. Previous reports suggest that plasma-mediated growth inhibition exhibits a certain degree of selectivity, being more pronounced in tumor cells than in normal cells, likely due to differences in intracellular ROS levels associated with higher proliferative activity in cancer cells.
Based on this background, we are exploring plasma-based cancer therapy using indirect irradiation, in which atmospheric pressure plasma is applied to culture medium to generate plasma-activated medium (PAM), which can then be administered intraperitoneally. This approach is particularly aimed at effectively suppressing intraperitoneal dissemination of ovarian cancer. We are also investigating the molecular mechanisms underlying the antitumor effects of plasma and its impact on epithelial–mesenchymal transition (EMT), a key process promoting ovarian cancer dissemination.
Expression and Functional Analysis of Glycosyltransferases GnT-IVa and C2GnT in Choriocarcinoma
It is known that hydatidiform moles can progress to invasive moles and ultimately to choriocarcinoma; however, the underlying mechanisms remain unclear. Human chorionic gonadotropin (hCG), secreted by all trophoblastic diseases and placental trophoblasts, is utilized as a tumor marker and possesses four potential N-linked and four O-linked glycosylation sites.
Compared with normal pregnancy and hydatidiform moles, excessively glycosylated hCG is markedly increased in invasive moles and choriocarcinoma. We are investigating the roles of glycosyltransferases responsible for this aberrant glycosylation, including N-acetylglucosaminyltransferase IV (GnT-IV) (Niimi K et al., British Journal of Cancer, 2012) and core-2 N-acetylglucosaminyltransferase-1 (C2GnT-1), which are associated with malignant transformation of trophoblastic cells.
Identification of the Causative Pregnancy in Gestational Trophoblastic Neoplasia
Gestational trophoblastic neoplasia includes invasive moles, choriocarcinoma, placental site trophoblastic tumors (PSTT), and epithelioid trophoblastic tumors (ETT). While invasive moles arise from hydatidiform moles, the causative pregnancy in other types of trophoblastic tumors is not necessarily the immediately preceding pregnancy. We investigate the causative pregnancy of gestational trophoblastic neoplasia using DNA analysis (Yamamoto E et al., Gynecologic Oncology Case Reports, 2014). Non-gestational choriocarcinoma can also be diagnosed using DNA-based methods.
Perinatal Research
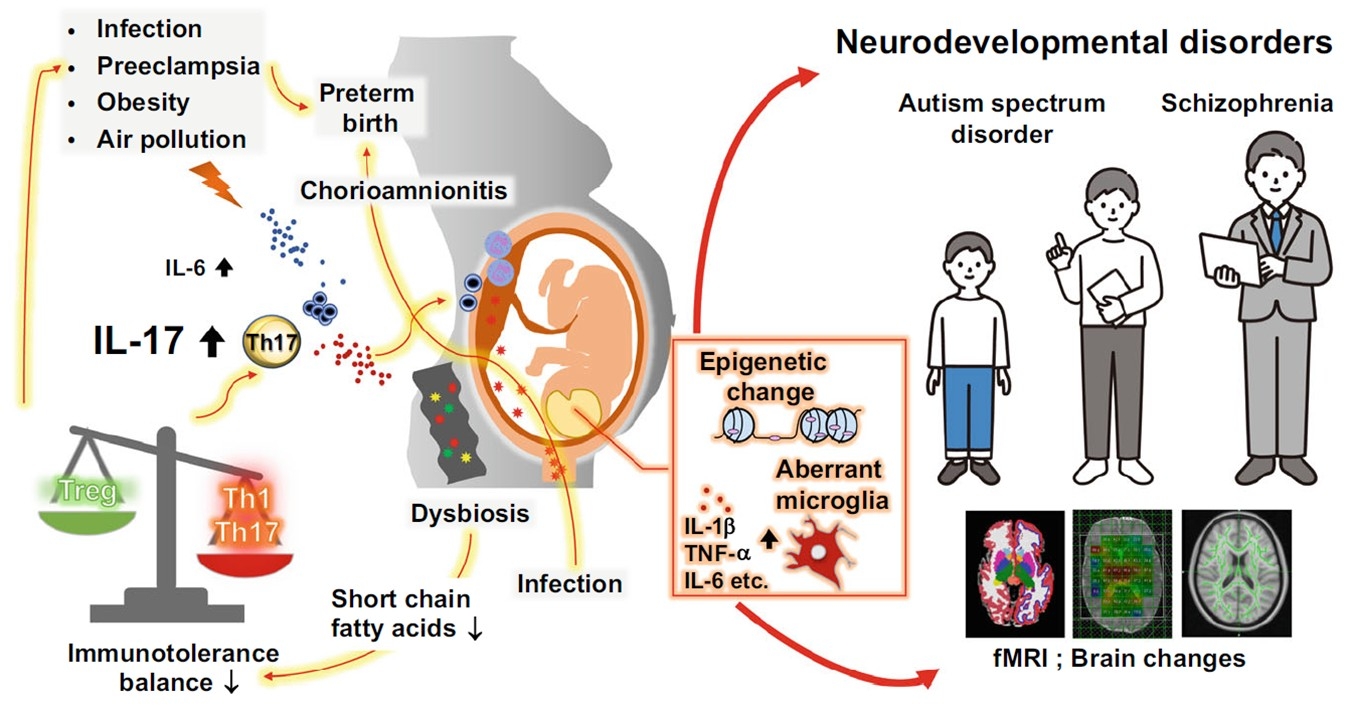
Impact of Hypertensive Disorders of Pregnancy and Intrauterine Environment on Child Development
Maternal hypertension, inflammation, medications, and nutrition during pregnancy may have long-term effects on fetal organ and brain development. In our group, clinical samples including maternal blood, cord blood, amniotic fluid, placenta, and umbilical cord are collected, stored, and analyzed after obtaining consent. In collaboration with pediatrics, we have analyzed neonatal brain MRI and demonstrated that hypertensive disorders of pregnancy, chorioamnionitis, and antenatal steroids may influence neonatal brain development (Ushida et al., Pregnancy Hypertension, 2021; Katsuki et al., Early Human Development, 2021; Nosaka et al., Archives of Gynecology and Obstetrics, 2024; Fuma et al., European Journal of Obstetrics & Gynecology and Reproductive Biology, 2024). Animal studies using maternal hypertension and maternal inflammation mouse models are also being conducted to examine the effects of intrauterine environment on offspring brain development (Imai et al., Free Radical Biology & Medicine, 2016; Fuma et al., Communications Biology, 2025).
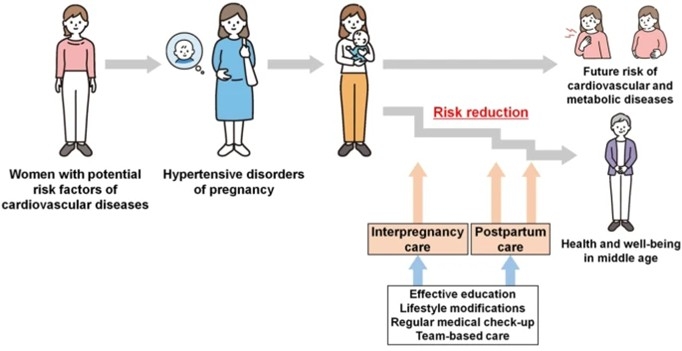
Prevention of Pregnancy Complications Through Lifestyle Modification Before and During Pregnancy
We are investigating whether lifestyle improvements—including weight management, diet, exercise, and sleep—can prevent pregnancy complications such as hypertensive disorders of pregnancy. Using clinical data collected from regional facilities and real-world data obtained from IoT devices, we aim to construct disease prevention algorithms incorporating patient education and apply them to our interconception care clinic (Ushida et al., Hypertension Research, 2024; Tano et al., Hypertension Research, 2024).
Identification of Factors Associated With Placenta Accreta Spectrum in Assisted Reproductive Technology
An increased incidence of placenta accreta spectrum has been reported in assisted reproductive technology, especially in frozen-thawed embryo transfer cycles with hormone replacement. Placenta accreta spectrum is associated with difficulty in placental separation after delivery and poses a high risk of massive postpartum hemorrhage. Because prenatal prediction is challenging, we conducted clinical studies to identify associated factors (Matsuo et al., Reproductive Medicine and Biology, 2024). Basic research on the pathophysiology of placenta accreta spectrum in hormone replacement cycles is currently underway.
Comprehensive EV-Focused Approaches for Improving Perinatal Outcomes
Extracellular vesicles released by all living cells carry proteins, nucleic acids, and lipids, and mediate intercellular communication. We previously demonstrated that prognosis of severe congenital diaphragmatic hernia—a life-threatening neonatal condition—can be predicted with high accuracy using amniotic fluid EVs (Matsuo et al. Journal of Extracellular Biology, 2024). We have recently initiated research on preeclampsia-related kidney injury.
Reproductive Endocrinology and Infertility Research
Elucidation of the Pathophysiology of Endometriosis and Development of Novel Therapeutic Approaches
Endometriosis is an estrogen-dependent chronic inflammatory disease, for which hormonal therapy remains the mainstay of treatment. However, hormonal therapies suppress ovulation and are therefore incompatible with patient’s desire to conceive. Moreover, some patients experience lesion progression or persistent pain despite hormonal therapy. Surgical excision of ovarian endometriomas has been shown to reduce anti-Müllerian hormone (AMH) levels, indicating damage to ovarian reserve.
To address these challenges, our department is pursuing research toward non-hormonal therapies that preserve ovarian function. We were the first in the world to establish a mouse model of ovarian endometriosis (Hayashi S et al., Redox Biology, 2020). Using this model, we demonstrated the efficacy of a non-hormonal therapy targeting the NLRP3 inflammasome without affecting eutopic endometrium (Murakami et al., Reproductive Biology and Endocrinology, 2022). We are also investigating cellular senescence as a novel characteristic of endometriosis and exploring senolytic therapies as potential new treatment strategies. Furthermore, based on the retrograde menstruation theory, we revealed that bacterial infection of the endometrium contributes to the development of endometriosis and that antibiotic treatment of infected endometrium may suppress disease onset (Muraoka A et al., Science Translational Medicine, 2023).
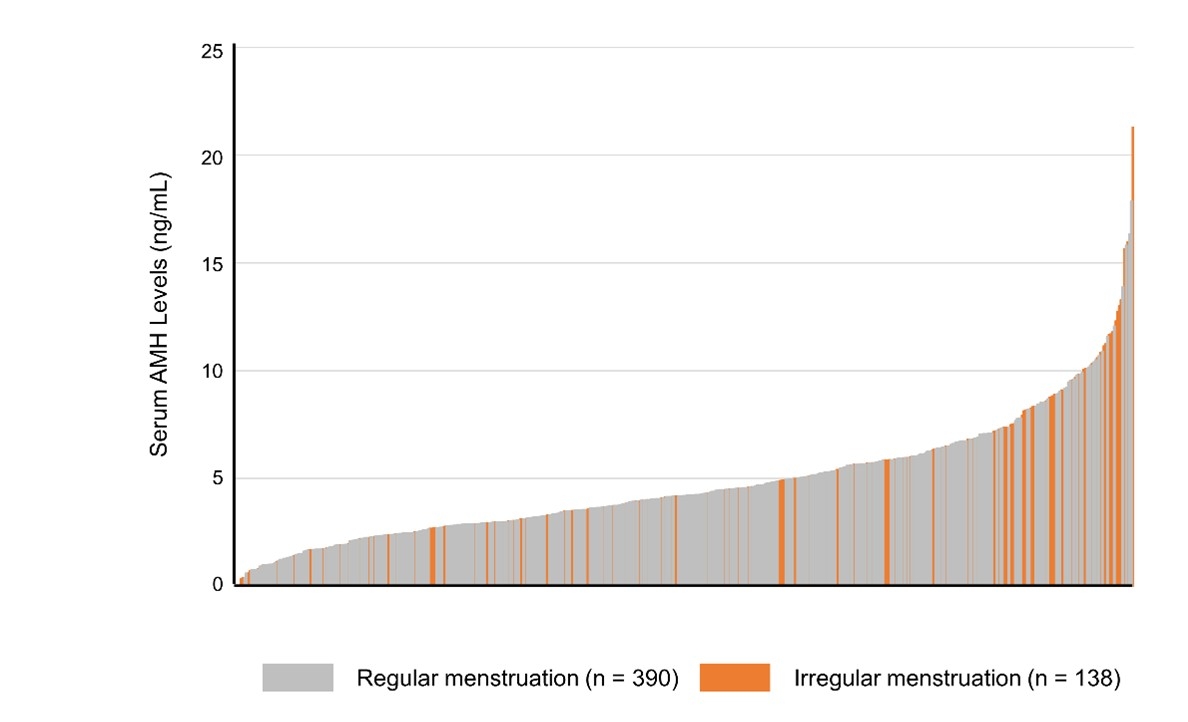
Exploration of Diagnosis and Pharmacotherapy for Polycystic Ovary Syndrome (PCOS) From a Preconception Care Perspective
PCOS is a common disorder affecting approximately 5–10% of women of reproductive age and is characterized by ovulatory dysfunction–related infertility, increased perinatal complications, and metabolic abnormalities such as obesity, diabetes, and hyperlipidemia. Despite its prevalence, the fundamental etiology remains unclear, and current treatments are mostly symptomatic treatments such as ovulation induction for ovulation disorder. In 2024, the Japan Society of Obstetrics and Gynecology revised the diagnostic criteria for PCOS to allow the use of serum AMH as a diagnostic marker. However, diagnostic approaches in adolescents and young women remain inconsistent. Having a simple method to identify the possibility of PCOS is critically important for young women who wish to conceive in the future. Our group investigated the relationship between serum AMH levels, iregular menstruation, and PCOS risk, using samples from several hundred female university students (Miyake N et al., Reproductive Medicine and Biology, 2024). Because PCOS is a systemic disorder involving multiple organs, elucidation of its pathophysiology requires multi-organ analysis. Given the difficulty of obtaining multiple human tissue samples simultaneously, appropriate animal models are essential. We successfully developed a PCOS-like animal model by prenatal maternal androgen exposure and demonstrated aberrant expression of kisspeptin—a hypothalamic neuropeptide regulating GnRH/LH secretion—as a contributor to elevated LH levels (Osuka S et al., Endocrinology, 2017). This model is currently being used in industry-academia collaborative studies to evaluate pharmacological treatments (Muraoka A et al., Science Translational Medicine, 2023).
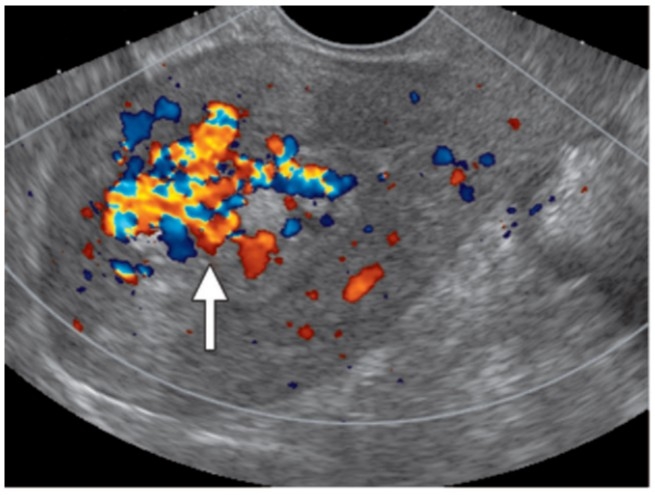
Prediction of Massive Hemorrhage in Retained Products of Conception (RPOC)
RPOC refers to retained chorionic tissue forming intrauterine masses after pregnancy termination and may cause life-threatening hemorrhage. Currently, no standardized treatment protocol exists. Establishing criteria to accurately identify cases requiring interventions such as uterine artery embolization or hysteroscopic surgery is essential. Our department identified predictors of massive hemorrhage in RPOC (Sonehara R et al., Scientific Reports, 2022) and is currently conducting a multicenter study to develop a hemorrhage prediction scoring system.
In addition to above, we are also conducting research on diagnostic biomarkers for premature ovarian insufficiency (POI) and mechanisms regulating follicular development using human granulosa cell lines, in collaboration with basic research laboratories across diverse fields.
BIBLIOGRAPHY
不明
- Yoshita S, Osuka S, Shimizu T, Fujitsuka N, Matsumoto C, Bayasula, Miyake N, Muraoka A, Nakanishi N, Nakamura T, Goto M, Kajiyama H. Unkeito promotes follicle development by restoring reduced follicle-stimulating hormone responsiveness in rats with polycystic ovary syndrome. [Journal name pending PubMed confirmation].
2025
- Muraoka A, Yokoi A, Yoshida K, Kitagawa M, Bayasula, Murakami M, Miyake N, Sonehara R, Nakamura T, Osuka S, Kajiyama H. Serum-derived small extracellular vesicles as biomarkers for predicting pregnancy and delivery in assisted reproductive technology in patients with endometriosis. Front Endocrinol (Lausanne). 2025;15:1442684.
- Fuma K, Iitani Y, Imai K, Ushida T, Tano S, Yoshida K, Yokoi A, Miki R, Kidokoro H, Sato Y, Hara Y, Ogi T, Nomaki K, Tsuda M, Komine O, Yamanaka K, Kajiyama H, Kotani T. Prenatal inflammation impairs early CD11c-positive microglia induction and delays myelination in neurodevelopmental disorders. Commun Biol. 2025 Jan 17;8(1):75. doi: 10.1038/s42003-025-07511-3.
- Tano S, Kotani T, Ushida T, Matsuo S, Yoshihara M, Imai K, Kinoshita F, Moriyama Y, Nomoto M, Yoshida S, Yamashita M, Kishigami Y, Oguchi H, Kajiyama H. Visualizing risk modification of hypertensive disorders of pregnancy: development and validation of prediction model for personalized interpregnancy weight management. Hypertens Res. 2025 Mar;48(3):884-893.
2024
- Yabuki A, Muraoka A, Tamauchi S, Seki T, Takeda T, Sonehara R, Miyake N, Nakamura T, Osuka S, Kajiyama H. The influence of radical trachelectomy on endometrial thickness in in vitro fertilization–embryo transfer. J Obstet Gynaecol Res. 2024;50(2):218–224.
- Yabuki A, Muraoka A, Osuka S, Yokoi A, Yoshida K, Kitagawa M, Bayasula, Sonehara R, Miyake N, Nakanishi N, Nakamura T, Iwase A, Kajiyama H. Serum miRNA as a predictive biomarker for ovarian reserve after endometrioma cystectomy. Reprod Biol. 2024;24(1):100821.
- Nosaka R, Ushida T, Kidokoro H, Kawaguchi M, Shiraki A, Iitani Y, Imai K, Nakamura N, Sato Y, Hayakawa M, Natsume J, Kajiyama H, Kotani T. Intrauterine exposure to chorioamnionitis and neuroanatomical alterations at term-equivalent age in preterm infants. Arch Gynecol Obstet. 2024 May;309(5):1909-1918.
- Fuma K, Ushida T, Kawaguchi M, Nosaka R, Kidokoro H, Tano S, Imai K, Sato Y, Hayakawa M, Kajiyama H, Kotani T. Impact of antenatal corticosteroids on subcortical volumes in preterm infants at term-equivalent age: A retrospective observational study. Eur J Obstet Gynecol Reprod Biol. 2024 Nov;302:7-14.
- Ushida T, Tano S, Imai K, Matsuo S, Kajiyama H, Kotani T. Postpartum and interpregnancy care of women with a history of hypertensive disorders of pregnancy. Hypertens Res. 2024 Jun;47(6):1457-1469.
- Miyake N, Osuka S, Ohsawa I, Tonoike T, Uno T, Tsuzuki K, Bayasula, Sonehara R, Muraoka A, Nakamura T, Goto M, Iwase A, Kajiyama H. Association between anti-Müllerian hormone levels and polycystic ovary syndrome in a general cohort of young women in Japan. Reprod Med Biol. 2024;23(1):e12615.
2023
- Muraoka A, Suzuki M, Hamaguchi T, Watanabe S, Iijima K, Murofushi Y, Shinjo K, Osuka S, Hariyama Y, Ito M, Ohno K, Kiyono T, Kyo S, Iwase A, Kikkawa F, Kajiyama H, Kondo Y. Fusobacterium infection facilitates the development of endometriosis through the phenotypic transition of endometrial fibroblasts. Sci Transl Med. 2023;15(700):eadd1531.
- Nakanishi N, Osuka S, Kono T, Kobayashi H, Ikeda S, Bayasula, Sonehara R, Murakami M, Yoshita S, Miyake N, Muraoka A, Kasahara Y, Murase T, Nakamura T, Goto M, Iwase A, Kajiyama H. Upregulated ribosomal pathway impairs follicle development in a polycystic ovary syndrome mouse model: differential gene expression analysis of oocytes. Reprod Sci. 2023;30(4):1306–1315.
2022
- Miyake N, Nagai T, Suga H, Osuka S, Kasai T, Sakakibara M, Soen M, Ozaki H, Miwata T, Asano T, Kano M, Muraoka A, Nakanishi N, Nakamura T, Goto M, Yasuda Y, Kawaguchi Y, Miyata T, Kobayashi T, Sugiyama M, Onoue T, Hagiwara D, Iwama S, Iwase A, Inoshita N, Arima H, Kajiyama H. Functional lactotrophs in induced adenohypophysis differentiated from human iPS cells. Endocrinology. 2022;163(3):bqac004.
- Wei W, Komatsu K, Osuka S, Murase T, Bayasula, Nakanishi N, Nakamura T, Goto M, Iwase A, Masubuchi S, Kajiyama H. Tamoxifen activates dormant primordial follicles in mouse ovaries. Reprod Sci. 2022;29(12):3404–3412.
- Murakami M, Osuka S, Muraoka A, Hayashi S, Bayasula, Kasahara Y, Sonehara R, Hariyama Y, Shinjo K, Tanaka H, Miyake N, Yoshita S, Nakanishi N, Nakamura T, Goto M, Kajiyama H. Effectiveness of NLRP3 inhibitor as a non-hormonal treatment for ovarian endometriosis. Reprod Biol Endocrinol. 2022;20(1):58.
- Sonehara R, Nakamura T, Iwase A, Nishida K, Takikawa S, Murakami M, Yoshita S, Muraoka A, Miyake N, Nakanishi N, Osuka S, Goto M, Kajiyama H. Predictive factors for massive hemorrhage in women with retained products of conception: a prospective study. Sci Rep. 2022;12(1):11859.
2021
- Yoshida K, Yokoi A, Sugiyama M, Oda S, Kitami K, Tamauchi S, Ikeda Y, Yoshikawa N, Nishino K, Niimi K, Suzuki S, Kikkawa F, Yokoi T, Kajiyama H. Expression of the chrXq27.3 miRNA cluster in recurrent ovarian clear cell carcinoma and its impact on cisplatin resistance. Oncogene. 2021 Feb;40(7):1255-1268.
- Ushida T, Kidokoro H, Nakamura N, Katsuki S, Imai K, Nakano-Kobayashi T, Moriyama Y, Sato Y, Hayakawa M, Natsume J, Kajiyama H, Kotani T. Impact of maternal hypertensive disorders of pregnancy on brain volumes at term-equivalent age in preterm infants: A voxel-based morphometry study. Pregnancy Hypertens. 2021 Aug;25:143-149. doi: 10.1016/j.preghy.2021.06.003. Epub 2021 Jun 9. PMID: 34139669.
- Katsuki S, Ushida T, Kidokoro H, Nakamura N, Iitani Y, Fuma K, Imai K, Nakano-Kobayashi T, Sato Y, Hayakawa M, Natsume J, Kajiyama H, Kotani T. Hypertensive disorders of pregnancy and alterations in brain metabolites in preterm infants: A multi-voxel proton MR spectroscopy study. Early Hum Dev. 2021 Dec;163:105479.
- Kasahara Y, Osuka S, Takasaki N, Bayasula, Koya Y, Nakanishi N, Murase T, Nakamura T, Goto M, Iwase A, Kajiyama H. Primate-specific POTE-actin gene could play a role in human folliculogenesis by controlling the proliferation of granulosa cells. Cell Death Discov. 2021;7(1):186.
- Muraoka A, Osuka S, Yabuki A, Bayasula, Yoshihara M, Tanaka H, Sonehara R, Miyake N, Murakami M, Yoshita S, Nakanishi N, Nakamura T, Goto M, Iwase A, Kajiyama H. Impact of perioperative use of GnRH agonist or dienogest on ovarian reserve after cystectomy for endometriomas: a randomized controlled trial. Reprod Biol Endocrinol. 2021;19(1):179.
2020
- Yasui H, Kajiyama H, Tamauchi S, Suzuki S, Peng Y, Yoshikawa N, Sugiyama M, Nakamura K, Kikkawa F. CCL2 secreted from cancer-associated mesothelial cells promotes peritoneal metastasis of ovarian cancer cells through the P38-MAPK pathway. Clin Exp Metastasis. 2020 Feb;37(1):145-158.
- Yoshihara M, Kajiyama H, Yokoi A, Sugiyama M, Koya Y, Yamakita Y, Liu W, Nakamura K, Moriyama Y, Yasui H, Suzuki S, Yamamoto Y, Ricciardelli C, Nawa A, Shibata K, Kikkawa F. Ovarian cancer-associated mesothelial cells induce acquired platinum-resistance in peritoneal metastasis via the FN1/Akt signaling pathway. Int J Cancer. 2020 Apr 15;146(8):2268-2280.
- Hayashi S, Nakamura T, Motooka Y, Ito F, Jiang L, Akatsuka S, Iwase A, Kajiyama H, Kikkawa F, Toyokuni S. Novel ovarian endometriosis model causes infertility via iron-mediated oxidative stress in mice. Redox Biol. 2020;37:101726.
- Muraoka A, Osuka S, Kiyono T, Suzuki M, Yokoi A, Murase T, Nishino K, Niimi K, Nakamura T, Goto M, Kajiyama H, Kondo Y, Kikkawa F. Establishment and characterization of cell lines from human endometrial epithelial and mesenchymal cells from patients with endometriosis. FS Sci. 2020;1(2):195–205.
2019
- Peng Y, Kajiyama H, Yuan H, Nakamura K, Yoshihara M, Yokoi A, Fujikake K, Yasui H, Yoshikawa N, Suzuki S, Senga T, Shibata K, Kikkawa F. PAI-1 secreted from metastatic ovarian cancer cells triggers the tumor-promoting role of the mesothelium in a feedback loop to accelerate peritoneal dissemination. Cancer Lett. 2019 Feb 1;442:181-192.
- Yokoi A, Villar-Prados A, Oliphint PA, Zhang J, Song X, De Hoff P, Morey R, Liu J, Roszik J, Clise-Dwyer K, Burks JK, O'Halloran TJ, Laurent LC, Sood AK. Mechanisms of nuclear content loading to exosomes. Sci Adv. 2019 Nov 20;5(11):eaax8849.
- Yoshida K, Yokoi A, Kagawa T, Oda S, Hattori S, Tamauchi S, Ikeda Y, Yoshikawa N, Nishino K, Utsumi F, Niimi K, Suzuki S, Shibata K, Kajiyama H, Yokoi T, Kikkawa F. Unique miRNA profiling of squamous cell carcinoma arising from ovarian mature teratoma: comprehensive miRNA sequence analysis of its molecular background. Carcinogenesis. 2019 Dec 31;40(12):1435-1444.
2018
- Fujikake K, Kajiyama H, Yoshihara M, Nishino K, Yoshikawa N, Utsumi F, Suzuki S, Niimi K, Sakata J, Mitsui H, Shibata K, Senga T, Kikkawa F. A novel mechanism of neovascularization in peritoneal dissemination via cancer-associated mesothelial cells affected by TGF-β derived from ovarian cancer. Oncol Rep. 2018 Jan;39(1):193-200.
2017
- Yokoi A, Yoshioka Y, Yamamoto Y, Ishikawa M, Ikeda SI, Kato T, Kiyono T, Takeshita F, Kajiyama H, Kikkawa F, Ochiya T. Malignant extracellular vesicles carrying MMP1 mRNA facilitate peritoneal dissemination in ovarian cancer. Nat Commun. 2017 Mar 6;8:14470.
- Osuka S, Iwase A, Nakahara T, Kondo M, Saito A, Bayasula, Nakamura T, Takikawa S, Goto M, Kotani T, Kikkawa F. Kisspeptin in the hypothalamus of two rat models of polycystic ovary syndrome. Endocrinology. 2017;158(2):367–377.
2016
- Imai K, Kotani T, Tsuda H, Mano Y, Nakano T, Ushida T, Li H, Miki R, Sumigama S, Iwase A, Hirakawa A, Ohno K, Toyokuni S, Takeuchi H, Mizuno T, Suzumura A, Kikkawa F. Neuroprotective potential of molecular hydrogen against perinatal brain injury via suppression of activated microglia. Free Radic Biol Med. 2016 Feb;91:154-63.
2014
- Yamamoto E, Niimi K, Shinjo K, Yamamoto T, Fukunaga M, Kikkawa F. Identification of causative pregnancy of gestational trophoblastic neoplasia diagnosed during pregnancy by short tandem repeat analysis. Gynecol Oncol Case Rep. 2014;9:3–6.
2012
- Niimi K, Yamamoto E, Fujiwara S, Shinjo K, Kotani T, Umezu T, Kajiyama H, Shibata K, Ino K, Kikkawa F. High expression of N-acetylglucosaminyltransferase IVa promotes invasion of choriocarcinoma. Br J Cancer. 2012;107(12):1969–1977.
MESSAGE
Call for graduate students
We welcome overseas students and doctors. Opportunities exist for students wishing to undertake postgraduate degrees in Ph.D.

