Target and Drug Discovery
KEYWORDS
- Cancer
- Signal Transduction
- EVs
- Rafts
HEAD

ONEYAMA Chitose
Adjunct Professor
CONTACT
| coneyama◎aichi-cc.jp(Please send a message after replacing "◎" mark with "@" mark. ) |
OUTLINE
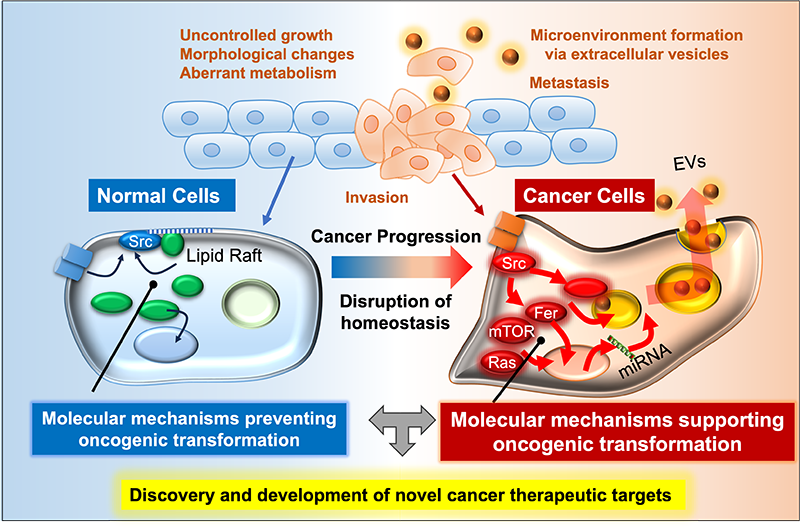
Our laboratory investigates the molecular mechanisms that maintain cellular homeostasis and how their disruption drives cancer development. Normal cells precisely regulate signaling pathways in space, time, and quantity. Genetic and epigenetic alterations, or abnormal extracellular cues, disturb these networks, leading to uncontrolled growth and tumor progression.
By dissecting key genes, proteins, and signaling cascades, we aim to uncover how oncogenic phenotypes arise and to identify novel therapeutic targets. Our ultimate goal is to translate these insights into innovative strategies for cancer treatment.
RESEARCH PROJECTS
1) Innovative Imaging and Mechanistic Insights into Exosome Secretion in Cancer
Extracellular vesicles (EVs), including exosomes, are nanoscale particles secreted by cells that mediate intercellular communication. In cancer, EV secretion is markedly increased and contributes to tumor progression by remodeling the microenvironment and establishing pre-metastatic niches. To accelerate research on exosome biogenesis, we developed a quantitative cell system expressing fusion proteins of exosome markers (CD9, CD63, CD81) with the high-intensity luminescent protein NanoLuc. In these cells, luminescence in the culture medium correlates with exosome number, enabling efficient measurement of exosome secretion (Sci Rep, 2018). By transplanting these cells into mice, we established an ex vivo system for long-term tracking of secreted exosomes under physiologically relevant conditions. Furthermore, by incorporating BRET-based red-shifted luminescence, we achieved non-invasive in vivo imaging of exosomes (Sci Rep, 2020; Methods Mol Biol, 2022). Using these systems, we uncovered key mechanisms of exosome secretion. We demonstrated that Src tyrosine kinase, localized on endosomal membranes, interacts specifically with the ESCRT-associated protein Alix, promoting intraluminal vesicle (ILV) formation—the precursor of exosomes. We also identified differences among Src family kinases in their ability to enhance exosome secretion, which correlated with their affinity for lipid rafts (Sci Rep, 2019). Additionally, we found that SNARE proteins, involved in membrane fusion, influence Src localization and exosome release (Biol Pharm Bull, 2022). Moreover, we discovered a strong correlation between MEK/ERK pathway activation and increased exosome secretion. Mechanistically, MEK activation downregulates multiple genes involved in endosomal degradation, suppressing lysosomal processing and leading to accumulation of late endosomes. Inhibition of MEK restored endosomal degradation and significantly reduced exosome secretion, which in turn suppressed oncogenic phenotypes. These findings suggest that in cancers with frequent Ras/Raf mutations, MEK activation promotes exosome secretion by impairing endosomal degradation, potentially serving as a compensatory mechanism for maintaining intracellular homeostasis disrupted during tumorigenesis (Cancer Sci, 2022).
Our findings provide powerful tools and insights for studying exosome dynamics in vivo and open new avenues for targeting exosome-mediated communication in cancer therapy. 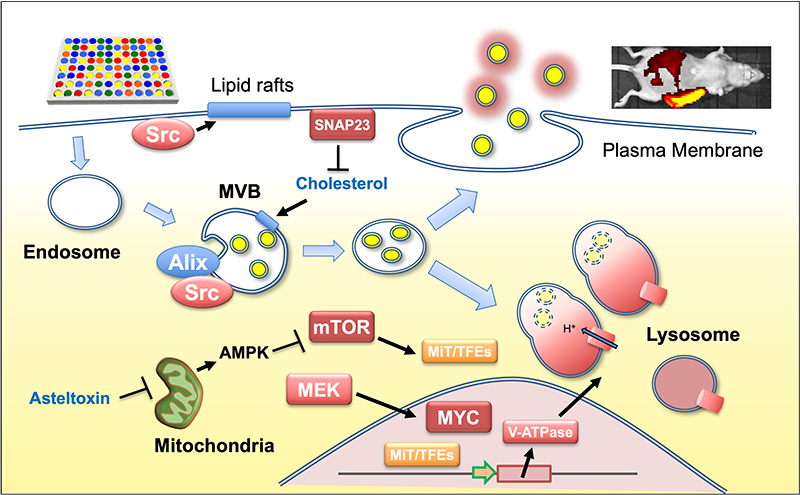
2) Spatial Regulation of Oncogenic Signaling and Development of Cancer Therapies Targeting Multimeric Tyrosine Kinases
The non-receptor tyrosine kinase c-Src is frequently upregulated in human cancers, driving malignant progression. However, the molecular circuits underlying Src oncogenic signaling remain poorly understood. Our studies revealed that Fer tyrosine kinase acts as a critical mediator and amplifier of Src-induced tumor progression (Oncogene, 2016). We previously demonstrated that fibroblast transformation is promoted by the relocation of c-Src from lipid rafts to non-raft membranes (Mol Cell, 2008; Mol Cell Biol, 2009). Under these conditions, Fer and ezrin were identified as non-raft Src targets. Mechanistically, c-Src initiates Fer autophosphorylation, which is further amplified through Fer oligomerization. Fer interacts with active Src at focal adhesions and phosphorylates ezrin, promoting cytoskeletal remodeling and cell transformation. Importantly, Fer activation is essential for tumorigenesis and invasiveness in cancers with elevated Src activity, including those driven by v-Src or EGFR signaling. Fer expression is upregulated in multiple cancers (colon, lung, pancreas, breast, kidney), yet its knockdown has minimal impact on normal cell growth, highlighting its potential as a selective therapeutic target.
To translate these findings, we screened small molecules targeting Fer and identified lead compounds that strongly inhibit cancer cell proliferation while exhibiting low toxicity toward normal cells. These candidates represent a new class of selective kinase inhibitors, distinct from conventional TKIs, and may offer highly specific cancer therapies.
Our work uncovers the Src–Fer axis as a novel oncogenic signaling pathway and provides a foundation for developing precision therapies targeting multimeric tyrosine kinases.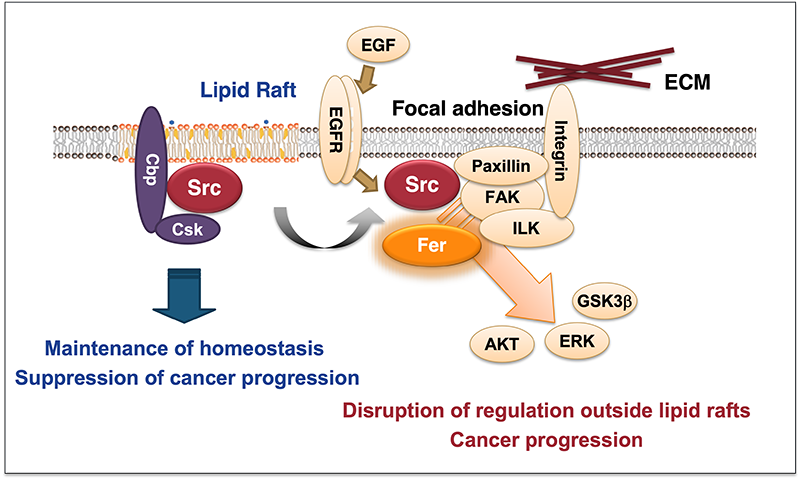
3) MicroRNA-Mediated Regulation of Oncogenic Signaling: Uncovering the Hidden Layers of Cancer Progression
The non-receptor tyrosine kinase c-Src is frequently overexpressed and activated in many human cancers, yet the molecular mechanisms driving its oncogenic effects remain unclear. To investigate this, we developed an experimental system using Csk-deficient mouse embryonic fibroblasts, where c-Src activation induces cellular transformation. Using this model, we compared microRNA (miRNA) expression profiles between non-transformed and c-Src-transformed cells. Microarray analysis revealed that c-Src activation alters a limited set of miRNAs: seven downregulated and six upregulated (>2-fold). Functional studies demonstrated that these miRNAs mediate c-Src-driven oncogenic signaling and create crosstalk with other pathways, including mTOR and focal adhesion signaling. Importantly, signaling molecules such as Src and mTOR are interconnected not only through phosphorylation and protein-protein interactions but also via translational regulation by miRNAs (Oncogene, 2011; Oncogene 2012, Cancer Sci, 2016, Cancer Sci, 2020). This interplay between kinases and miRNAs provides new insights into cancer progression, invasiveness, and drug resistance. Understanding miRNA-mediated regulation of Src signaling may lead to novel therapeutic strategies targeting these non-coding RNAs.
Our findings highlight miRNAs as critical regulators of oncogenic signaling networks and potential targets for precision cancer therapy. 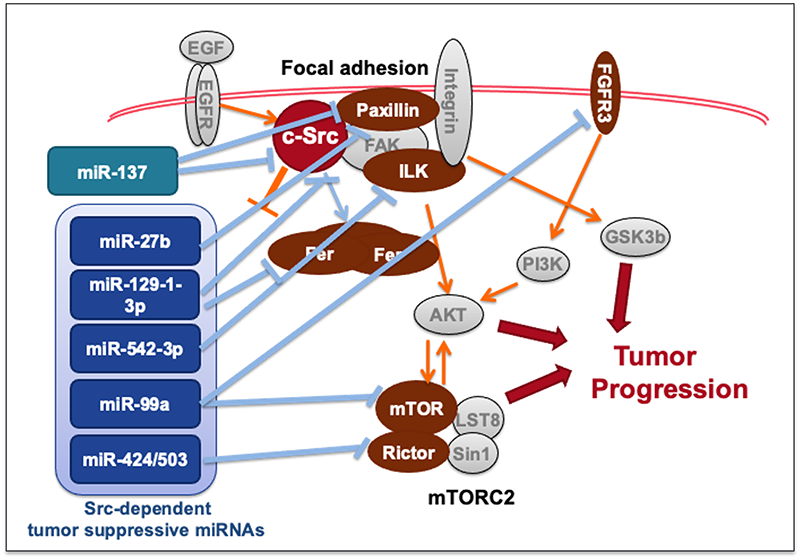
BIBLIOGRAPHY
2024
- Kunitake K, Mizuno T, Hattori K, Oneyama C, Kamiya M, Ota S, Urano Y, Kojima R. Barcoding of small extracellular vesicles with CRISPR-gRNA enables comprehensive, subpopulation-specific analysis of their biogenesis/release regulators. Nat Commun. 15(1), 9777 (2024)
2022
- Mitani F, Lin J, Sakamoto T, Uehara R, Hikita T, Yoshida T, Setiawan A, Arai M, Oneyama C. Asteltoxin inhibits extracellular vesicle production through AMPK/mTOR-mediated activation of lysosome function. Scientific Reports, 12(1), 6674 (2022)
- Hikita T, Uehara R, Itoh RE, Mitani F, Miyata M, Yoshida T, Yamaguchi R, Oneyama C. MEK/ERK-mediated oncogenic signals promote secretion of extracellular vesicles by controlling lysosome function. Cancer Sci, 113(4), 1264-1276 (2022)
- Mitani F, Hayasaka R, Hirayama A, Oneyama C. SNAP23-mediated perturbation of cholesterol-enriched membrane microdomain promotes extracellular vesicle production in Src-activated cancer cells. Biol Pharm Bull, 45(10), 1572-1580 (2022)
- Hikita T, Oneyama C. Quantification and imaging of exosomes via luciferase-fused exosome marker proteins: ExoLuc system. Methods Mol Biol, 2524, 281-290 (2022)
- Morioka S, Nakanishi H, Yamamoto T, Hasegawa J, Tokuda E, Hikita T, Sakihara T, Zhang Y, Oneyama C, Yamazaki M, Suzuki A, Sasaki J, Sasaki T. A novel comprehensive method for measuring the regioisomers of phosphoinositides. Nat Commun, 13(1),83 (2022)
2021
- Nishimura T, Oyama T, Hu HT, Fujioka T, Hanawa-Suetsugu K, Ikeda K, Yamada S, Kawana H, Saigusa D, Ikeda H, Kurata R, Oono-Yakura K, Kitamata M, Kida K, Hikita T, Mizutani K, Yasuhara K, Mimori-Kiyosue Y, Oneyama C, Kurimoto K, Hosokawa Y, Aoki J, Takai Y, Arita M, Suetsugu S. Filopodium-derived vesicles produced by MIM enhance the migration of recipient cells. Dev Cell, 56(6), 842-859 (2021)
2020
- Okuzaki D, Yamauchi T, Mitani F, Miyata M, Ninomiya Y, Watanabe R, Akamatsu H, Oneyama C.: c-Src promotes tumor progression via downregulation of miR-129-1-3p. Cancer Science, 111(2): 418-428 (2020)
- Hikita T, Miyata M, Watanabe R, Oneyama C. In vivo imaging of long-term accumulation of cancer-derived exosomes using a BRET-based reporter. Scientific Reports, 10(1):16616 (2020)
2019
- Hikita T, Kuwahara A, Watanabe R, Miyata M, Oneyama C. Src in endosomal membranes promotes exosome secretion and tumor progression. Scientific Reports, 9(1):3265 (2019)
MESSAGE
We are currently seeking graduate students to join our research team. Each experiment we conduct is a step toward new discoveries, and we hope to share the excitement and joy of that journey together.
If you are interested, please feel free to contact us.

