Advanced Cancer Therapeutics
KEYWORDS
- KRAS
- Resistance Mechanisms
- BRAF
- Target therapy
- ctDNA
HEAD

EBI Hiromichi
Adjunct Professor
CONTACT
| iga-ryu◎t.mail.nagoya-u.ac.jp (Please send a message after replacing "◎" mark with "@" mark. ) | |
| HP | Private Page |
OUTLINE
We focus on developing treatments for cancers that harbor abnormalities in the KRAS and BRAF genes, which are present in approximately 40% of all cancers. For BRAF V600E mutant colorectal cancer, based on research led by our laboratory, combination therapy with BRAF inhibitors and anti-EGFR antibody drugs has become the standard clinical treatment strategy for patients with BRAF mutations. Our findings are also used as reference data when determining treatment strategies for various BRAF mutations. For cancers with KRAS gene mutations, as the development of inhibitors has rapidly advanced in recent years, we are particularly dedicated to elucidating mechanisms of drug resistance and proposing novel therapeutic approaches to overcome such resistance.
RESEARCH PROJECTS
i) Elucidating Mechanisms of Resistance to KRAS Inhibitors and Developing Novel Therapies
The KRAS gene is frequently mutated in lung cancer (5%), colorectal cancer (40%), and pancreatic cancer (over 80%), and plays a critical role in cancer cell survival and proliferation. For 40 years since its discovery, no effective drugs were available, and KRAS was considered “undruggable.” However, in 2022, a KRAS-targeted drug was approved in Japan, and research in this area is now rapidly advancing. Our laboratory has demonstrated that the efficacy of KRAS inhibitors is associated with the epithelial-to-mesenchymal transition (EMT), and that KRAS-mutant tumors undergoing EMT exhibit reduced dependency on KRAS, rendering them insensitive to KRAS inhibitors (CCR 2020). Furthermore, we revealed that within 24 hours of KRAS inhibitor exposure, tumor cells induce changes in the localization of membrane proteins involved in maintaining epithelial characteristics, which in turn activates YAP signaling. Activation of YAP signaling leads to the induction of MRAS, a member of the RAS superfamily, allowing MAPK activity to be maintained even when KRAS is suppressed (Nature Cancer 2023). Additionally, we discovered that KRAS inhibitor treatment induces the expression of CD47—a “don’t eat me” signal that inhibits macrophage-mediated phagocytosis—thereby contributing to resistance to both KRAS inhibitors and immune checkpoint inhibitors (Cell Reports Medicine 2025).
ii) Development of Personalized Therapies for BRAF-Mutant Tumors
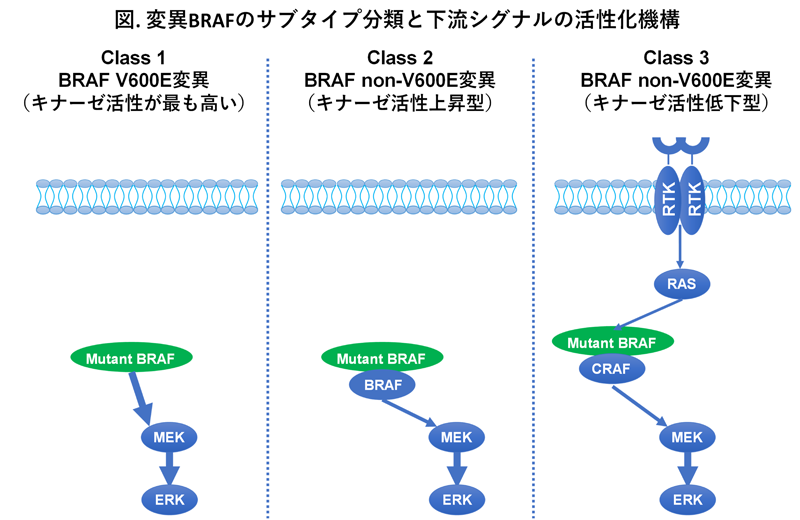
BRAF gene mutations represent a driver oncogenic alteration found in approximately 8% of all solid tumors, occurring in about 50% of melanomas, 30–70% of thyroid cancers, 10% of colorectal cancers, and 1% of lung cancers. Most mutations occur at the 600th amino acid residue, valine (BRAF V600). However, with the advancement of next-generation sequencing, including gene panel testing, it has become clear that non-V600 mutations account for roughly one-third of BRAF mutations in colorectal cancer and about half in lung cancer.
BRAF mutations are classified into three subtypes based on the kinase activity of the mutant BRAF protein. Class 1 consists of BRAF V600 mutations, where kinase activity is increased approximately 500-fold compared to wild-type BRAF, and the mutant BRAF monomer directly activates downstream signaling. In contrast, non-V600 mutations include Class 2, where kinase activity is moderately increased (several-fold to ~50-fold), and Class 3, where kinase activity is decreased. Class 2 mutant BRAF forms dimers with wild-type BRAF to activate downstream signaling. Class 3 mutant BRAF, despite reduced kinase activity, forms dimers with wild-type BRAF or CRAF, and these dimers are activated by receptor tyrosine kinase (RTK)-RAS signaling, thereby activating downstream pathways (see figure).
BIBLIOGRAPHY
2025
- Hirade K, Tanaka N, Kajino T, Adachi Y, Kimura R, Kasuya H, Kisoda S, Tan TK, Hayakawa S, Sato T, Yanase S, Kitaura Y, Yamamoto T, Nishioka Y, Muto O, Muraoka D, Fujishita T, Kasuga N, Watanabe K, Sakata Y, Aoki M, Matsushita H, Sanda T, Iida S, Tsuchiya K, Yamaguchi R, Ebi H. Inhibiting KRAS with CD47 and immune checkpoint overcomes intrinsic resistance to combined KRAS and immune checkpoint inhibitor therapy. Cell Rep Med. 6:102317, 2025
2023
- Adachi Y, Kimura R, Hirade K, Yanase S, Nishioka Y, Kasuga N, Yamaguchi R, Ebi H. Scribble mis-localization induces adaptive resistance to KRAS G12C inhibitors through feedback activation of MAPK signaling mediated by YAP-induced MRAS. Nat Cancer. 4:829-843, 2023.
2020
- Adachi Y, Ito K, Hayashi Y, Kimura R, Tan TZ, Yamaguchi R, Ebi H. Epithelial-to-Mesenchymal Transition is a Cause of Both Intrinsic and Acquired Resistance to KRAS G12C Inhibitor in KRAS G12C-Mutant Non-Small Cell Lung Cancer. Clin Cancer Res. 26:5962-5973, 2020.
2019
- Yaeger R, Kotani D, Mondaca S, Parikh, AR, Bando H, Van Seventer EE, Taniguchi H, Zhao H, Thant CN, de Stanchina E, Rosen N, Corcoran RB, Yoshino T, Yao Z, Ebi H. Response to Anti-EGFR Therapy in Patients with BRAF non-V600-Mutant Metastatic Colorectal Cancer. Clin Cancer Res 2019.
MESSAGE
KRAS inhibitors have become one of the major topics in cancer therapy. Our laboratory has a strong track record of collaboration and exchange with RAS researchers worldwide, and we welcome the participation of those interested in novel therapeutic development and the mechanisms of drug action.

